Weekly Roundup -
March 4, 2026
Smart. Strategic. Essential.
Unmatched Healthcare Insights from HMA,
Leavitt Partners & Wakely.
Featured:
Proposed ACA 2027 Notice of Benefit and Payment Parameters: Implications for Issuers and States
ACCESS WEBINARTrending: In Focus
The Value Shift in Medicare Advantage: What 2026 Benefits Tell Us About the Market’s Next Chapter
The Medicare Advantage (MA) program continues to evolve as plans respond to shifting policy signals, market pressures, and beneficiary expectations. A new paper from Wakely, an HMA Company—The Value Shift: How Medicare Advantage Benefits Are Evolving for 2026—provides a data-driven examination of how MA benefit designs are changing and what those changes signal about the future direction of the program.
This paper refreshes Wakely’s ongoing MA benefit analysis, updating prior findings with the latest 2026 plan enrollment data. It builds on Wakely’s established work examining benefit design, supplemental offerings, and the relationship between bids, rebates, and plan value, including The Value Shift: Inside the C-SNP Surge.
This article highlights findings from the proprietary value-add metric that Wakely developed to provide a comprehensive assessment of MA plan value. Although it can be used as a comparative metric to evaluate relative changes year over year, it is not intended to represent pricing.
From Benefit Expansion to Optimization
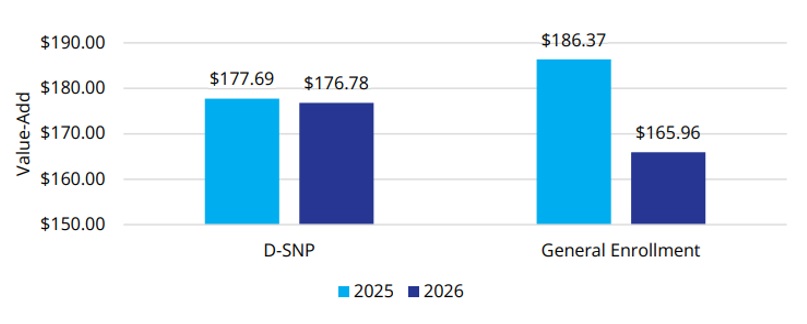
Over the past decade, MA plans have steadily expanded benefit offerings, supported by strong enrollment growth and favorable rebate dynamics. The 2026 benefit landscape suggests that plans have been taking a more measured approach (see Figure 1). Wakely’s analysis finds that plans are becoming more strategic in how benefits are designed and deployed, maintaining or enhancing benefits that are best aligned with quality performance, affordability, and target populations while pulling back in other areas.
Plans appear to be optimizing benefits to better align with member needs, quality performance, and financial parameters. Examples include refining supplemental benefits, adjusting cost-sharing structures, and rethinking how benefits support care management and health outcomes.
Figure 1. Change in Plan Value-Add from 2025 to 2026
The shift reflects an MA market in which differentiation and long-term sustainability are increasingly important.
Supplemental Benefits: More Targeted, More Strategic
Supplemental benefits remain a defining feature of Medicare Advantage, but their role is evolving. Wakely’s paper highlights a move away from expanding the number of benefits toward targeted benefit offerings that are more clearly connected to member engagement and outcomes.
Plans are homing their focus on benefits that support daily living, chronic condition management, and access to care, particularly for populations with higher needs. This targeted approach suggests plans are thinking about value, operational complexity, and how benefits contribute to overall value propositions.
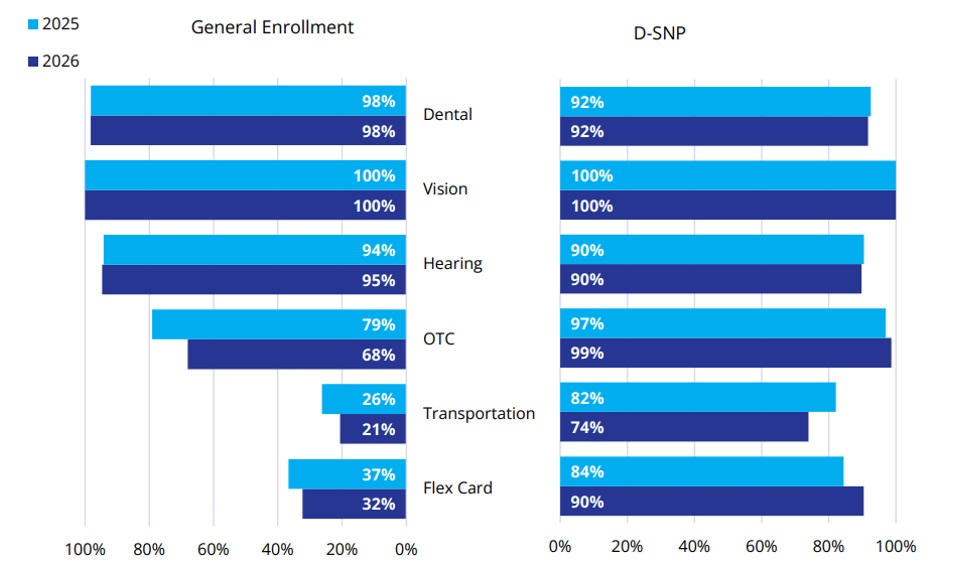
Between 2025 and 2026, the percentage of members with access to common supplemental benefits has, on average, stayed consistent or slightly decreased among the general enrollment population (Figure 2). The percentage of members who are enrolled in plans that offer over-the-counter (OTC) drug, transportation, and Flex Card benefits has decreased by 11 percent, 6 percent, and 4 percent, respectively. Conversely, the Dual Eligible Special Needs Plan (D-SNP) population saw an increase in member access to all supplemental benefit categories except transportation (an 8% decrease).
Figure 2. Percent of Enrollment in Common Supplemental Benefits
For stakeholders across the healthcare ecosystem, this trend underscores the importance of understanding not just what benefits are offered, but why.
Shifts in Cost Sharing and the Enrollee Experience
Wakely’s analysis also points to notable shifts in cost sharing and premium structures. There is continued attention to balancing affordability for members with the need to manage plan liability amid changing benchmarks and utilization patterns.
These decisions directly affect the member experience. Small shifts in copays, deductibles, or benefit limits can influence enrollment, retention, and satisfaction, particularly in competitive markets. As plans fine tune these levers, data-driven insights become critical to understanding how benefit changes may resonate with different member segments.
2026 Signals for Future Bid Cycles
The benefit trends identified in “The Value Shift” series suggest several broader signals for the MA market:
- Value over volume: Plans are prioritizing benefits that support quality, outcomes, and sustainable growth.
- Greater segmentation: Benefit designs are increasingly tailored to specific populations and market dynamics.
- Data-informed decision-making: As margins tighten, plans are relying more heavily on analytics to guide benefit strategy.
- Special needs plans continue to drive growth. Enrollment in Chronic Condition Special Needs Plans (C-SNPs) is the fastest-growing segment in MA.
These dynamics have implications for MA organizations and for providers, policymakers, and partners seeking to understand how MA continues to shape care delivery and costs.
Value-Add Metric and Benefit Design Insights
In this paper, Wakely paired its actuarial and analytic expertise with tools that enable detailed benefit and market analysis. One of those tools, Wakely’s Medicare Advantage Competitive Analysis Tool (WMACAT), calculates a comprehensive value-add metric that integrates five core components into a consistent framework that allows for apples-to-apples comparisons across plans, markets, and years. In addition, Wakely’s Strategic Market Analysis and Ranking Tool (SMART) supports broader competitive assessments by layering enrollment weighting, geographic variation, and plan positioning into the analysis.
As an HMA company, Wakely’s work is complemented by broader policy, market, and strategy expertise, helping organizations connect benefit decisions to regulatory developments, operational considerations, and long-term goals.
For health plans and healthcare organizations navigating the next phase of Medicare Advantage, these combined capabilities can respond to questions such as:
- How competitive is our benefit design today, and where are the risks?
- Which benefits are most aligned with our population and quality strategy?
- How might future policy or payment changes affect benefit sustainability?
Looking Ahead
MA benefit design remains an important signal of market direction by showing how plans are responding to policy change, market competition, and financial pressure. As plans shift from broad expansion to more targeted value strategies, the ability to measure, compare, and interpret benefit changes becomes essential as plans look ahead to the 2027 and 2028 bid cycles.
Wakely will continue to build on this work with upcoming analyses, including deeper dives into Part D design changes and the implications of the sunset of the Value-Based Insurance Design (VBID) program.
For information about this analysis and the Wakely tools, contact Dani Marino and Amanda Nelessen.
Federal Policy News
Fueled By Leavitt Partners Weekly Health Intelligence
Vaccine Policy Debate Intensifies as Surgeon General Nomination Advances
Although absent from last week’s State of the Union address, the Make America Health Again (MAHA) agenda, particularly with respect to vaccine policy, is continuing to have a significant influence on the US Department of Health and Human Services (HHS) and Congress. Last week, during the Senate HELP Committee’s nomination hearing for Surgeon General nominee, Dr. Casey Means, HHS Secretary Robert F. Kennedy’s recent actions impacting vaccine policy, in addition to the witness’s own perspective on vaccines, were a focal point for both Republicans and Democrats. Chair Bill Cassidy (R-LA), along with other majority members of the committee, was generally supportive of Dr. Means’ chronic disease and nutrition focus, but expressed concern regarding her responses to vaccine-related questions, including hepatitis B universal immunization recommendations, the MMR vaccine, flu vaccine recommendations, and the concept of “shared clinical decision-making,” which she encouraged in her responses to these questions. Chair Cassidy underscored concerns regarding increasing vaccine skepticism in the U.S., citing data that the ongoing measles outbreak in the U.S. has surpassed 1,000 cases in 2026 alone, and that the U.S. is at risk of losing its measles elimination status. In addition to the data cited by the Senator, recent modeling from the Yale School of Public Health, found that an annual decline in measles, mumps, and rubella (MMR) vaccine coverage of 1 percent over the next five years could lead to more than 17,000 measles cases, 4,000 hospitalizations, and 36 preventable deaths per year, costing $1.5 billion annually.
While a markup to vote on the nomination has not yet been scheduled, a simple majority is needed to advance the nomination of Dr. Means out of committee.
Meanwhile, on February 27, Secretary Kennedy announced two new members of the Advisory Committee on Immunization Practices (ACIP) ahead of its upcoming meeting on March 18 and 19. The two new members are Dr. Sean Downing, a practicing primary care physician, and Dr. Angelina Farella, a practicing pediatrician. According to statements from Secretary Kennedy and new Acting Centers for Disease Control and Prevention (CDC) Director Jay Bhattacharya in the HHS press release announcing the appointments, the new members are expected to provide the perspectives of practicing physicians who implement the recommendations made by ACIP. Notably, Dr. Farella had been outspoken against the COVID-19 vaccine, reportedly having testified in front of the Texas Senate that the vaccine should be removed from the market. The March ACIP meeting agenda includes discussion of COVID-19 vaccine injuries, long-COVID, and ACIP recommendation methodology. The meeting may also include recommendation votes related to COVID19 vaccine injuries and long-COVID, as well as ACIP recommendation methodology. Public comments for the rescheduled ACIP meeting will be accepted from March 2–12, 2026.
Separately, the Vaccines and Related Biological Products Advisory Committee (VRBPAC), which advises the US Food and Drug Administration (FDA), announced its next session will take place on March 12, 2026. VRBPAC is slated to discuss and make recommendations on the strain composition of influenza virus vaccines for use in the United States during the 2026–2027 influenza season.
CMS Defers Medicaid Funds, Imposes DME Moratorium in Anti-Fraud Push
On February 25, HHS and the Centers for Medicare & Medicaid Services (CMS) announced a three-part “crackdown” on Medicare and Medicaid fraud. The announced actions include deferring $259.5 million in quarterly federal Medicaid matching funds to Minnesota while CMS completes a program-integrity review in the state. Specifically, CMS pointed to $243.8 million in unsupported and potentially fraudulent claims, as well as $15.4 million tied to Medicaid claims for individuals lacking satisfactory immigration status. CMS also flagged what it characterized to be unusually rapid growth in personal care, home- and community-based services, and other practitioner spending in the state. CMS noted the state can respond with additional information and documentation during the review, but the deferrals in federal funding could increase if deficiencies persist.
Second, CMS announced a six-month nationwide moratorium effective February 27 on new Medicare enrollment for certain Durable Medical Equipment, Prosthetic Devices, Prosthetics, Orthotics, & Supplies (DMEPOS) suppliers, due to concerns regarding the prevalence of fraud within the spending area. The agency identified $1.5 billion in suspected fraudulent DMEPOS billing last year.
Finally, CMS announced it is seeking stakeholder input to inform development of future potential rulemaking under the Comprehensive Regulations to Uncover Suspicious Healthcare (CRUSH) initiative. In the CRUSH RFI, CMS seeks stakeholder input on additional anti-fraud tools and potential regulatory changes across Medicare, Medicaid, CHIP, and the Marketplace. The RFI specifically requests information on several possible anti-fraud measures, including “enhanced identity proofing and ownership requirements” (for Medicare-enrolled providers or suppliers), using AI to assist with Medicare Advantage coding, and opportunities to support state Medicaid program integrity. The RFI is open for public comment until March 30.
Education Department Shifts School Safety Programs to HHS
On February 23, the Department of Education (ED) announced a new interagency agreement (IAA) with HHS to transfer management of several ED programs focused on school safety and emergency preparedness to HHS as a part of the Trump Administration’s efforts to downsize the ED. It was previously announced in November that HHS would now manage the National Committee on Foreign Medical Education and Accreditation (NCFMEA) and Child Care Access Means Parents in School (CCAMPIS) Partnership, previously administered by ED. HHS will now also administer the School Emergency Response to Violence (Project SERV), School Safety National Activities, Ready to Learn Programming, Full-Service Community Schools, Promise Neighborhoods, and Statewide Family Engagement Centers programs. While no specific administrative, funding, or staffing changes for HHS were announced, this partnership will make HHS responsible for managing funding competitions and providing technical assistance to several initiatives regarding school safety, while ED will provide “proper oversight.”
HELP Committee Advances Living Donor and Cybersecurity Bills
On February 26, the Senate HELP Committee voted to advance two key healthcare bills out of committee. The first health-related bill, S. 1152, Living Donor Protection Act of 2025, was introduced by Senators Kirsten Gillibrand (D-NY) and Tom Cotton (R-AR) and agreed to by a vote of 22-1. The bill clarifies eligibility for time off under the Family Medical Leave Act (FMLA) and prohibits insurers from discriminating against living donors in life, disability, and long-term care insurance. The committee also advanced S. 3315, Health Care Cybersecurity and Resiliency Act of 2025, sponsored by Chair Bill Cassidy (R-LA) and Senators Maggie Hassan (D-NH), John Cornyn (R-TX), and Mark Warner (D-VA). The bill seeks to strengthen the healthcare sector’s preparedness against cyberattacks and was agreed to by a vote of 22-1. The House version of the Living Donor Protection Act, H.R. 4583, has not yet been advanced by the House Energy and Commerce Committee, while the Health Care Cybersecurity and Resiliency Act does not have a House companion.
Ready to talk about your organization's challenges?
Schedule a ConsultationState Policy News
Alaska
Alaska Opens Letter of Interest Portal for RHTP Funding. The Alaska Department of Health announced on February 18, 2026, that it has opened its Letter of Interest (LOI) portal for organizations interested in participating in the state’s implementation of the federal Rural Health Transformation Program. Organizations interested in RHTP funding can submit a LOI with project proposals aligning with one of Alaska’s RHTP initiatives: maternal and child health, expanding access to essential services closer to home, promoting healthy communities and preventative care, advancing value-based care and fiscal sustainability, strengthening the health care workforce, and modernizing technology infrastructure. Responses are due March 11, 2026, and funding awards are expected to begin in spring 2026.
Florida
Florida Lowers AIDS Drug Assistance Program Eligibility; CMS to Hold Marketplace Special Enrollment Period. This month the Florida Department of Health (DOH) issued an emergency rule that lowered eligibility for the AIDS Drug Assistance Program (ADAP) to 130 percent of the federal poverty level and limited insurance coverage of a once-daily pill that treats HIV used by 60 percent of people in ADAP, effective for 90 days beginning March 1, 2026. DOH cited funding concerns as the reason for the cuts. Previously, individuals making up to 400 percent of the federal poverty level could enroll in ADAP, which provides free FDA-approved HIV medication to eligible low-income, uninsured, or underinsured individuals. Approximately 30,000 individuals were enrolled in the program before the emergency rule and advocates estimate that up to 16,000 could be disenrolled. The Centers for Medicare & Medicaid Services (CMS) opened a special enrollment period for affected individuals to choose a new Marketplace plan by April 30, 2026.
Illinois
Illinois to Release Specialty Managed Care Plan for Foster Care Youth RFP in Summer 2026. The Illinois Department of Children and Family Services (DCFS) announced in February 2026 that it anticipates releasing a request for proposals (RFP) for the Specialty Managed Care Plan for Youth in Care and Former Youth in Care in late summer 2026. DCFS is holding a stakeholder listening session on March 5, 2026, allowing public comments to inform the procurement process on how care should be delivered in the program, which is currently called YouthCare. The department will begin its reprocurement process in spring 2026, and the new contract is anticipated to begin in January 2028. Centene is the incumbent YouthCare contractor.
Iowa
Iowa Issues RHTP Rural Workforce Recruitment, Medical Equipment RFP. The Iowa Department of Health and Human Services released two requests for proposals (RFPs) under the state’s Rural Health Transformation Program (RHTP) aimed at strengthening rural healthcare capacity. The first RFP focuses on recruiting healthcare providers to practice in rural communities through incentives such as recruitment bonuses, relocation assistance, and other financial supports for full-time providers. The contract runs from June 1, 2026, to September 30, 2031, and is worth $5 million. The second RFP seeks applicants to procure and install advanced medical equipment, such as CT scanners, radiotherapy units, and robotic surgical systems, to expand access to specialty services in rural areas. Iowa anticipates awarding approximately $50 million across roughly 25 awards for the medical equipment initiative. Applications for both opportunities are due April 9, 2026, with awards expected to be announced on or around May 22, 2026.
Minnesota
Minnesota Files Lawsuit to Challenge Federal Withholding of Medicaid Funds. On March 2, 2026, the Minnesota Attorney General’s office announced it has filed a federal lawsuit seeking to block the Trump administration’s withholding of approximately $243 million in Medicaid funding following the federal government’s decision to temporarily pause funding due to its fraud concerns. The state argues the action, which represents about 7 percent of its quarterly Medicaid allocation, was implemented without adequate procedural justification or formal findings of noncompliance. The administration has described the pause as part of broader fraud oversight efforts. The lawsuit names the US Department of Health and Human Services and the Centers for Medicare & Medicaid Services, as well as their respective leaders, HHS Secretary Robert F. Kennedy Jr. and CMS Administrator Dr. Mehmet Oz, in their official capacities.
North Carolina
North Carolina Medicaid Director Jay Ludlam Steps Down. In February, North Carolina Medicaid announced that Medicaid Director Jay Ludlam is stepping down. Assistant secretary of Medicaid Melanie Bush was named interim director. Ludlam is slated to become Plan President with Molina Healthcare.
Private Market News
Fueled By Wakely Consulting Group
BrightSpring Closes $238.5 Million Deal to Acquire UnitedHealth Home Health, Hospice Facilities
BrightSpring Health Services has closed a $238.5 million deal to acquire 107 home health and hospice facilities from UnitedHealth Group. The deal was a part of an antitrust settlement with the Department of Justice that required United to sell some of its home health locations to acquire the provider Amedisys for $3.3 billion.
Our Insights
Fueled By Experts Across Our HMA Companies
Health Management Associates
Webinar: Proposed ACA 2027 Notice of Benefit and Payment Parameters: Implications for Issuers and States
This webinar offers a timely, strategic overview of the recently released proposed 2027 Notice of Benefit and Payment Parameters and what it signals for the evolving coverage landscape. We will unpack the key policy changes, operational requirements, and market assumptions embedded in the rule, with a focus on implications for the individual and small group markets beginning in 2026 and extending beyond.
Wakely
The Value Shift – How Medicare Advantage Benefits Are Evolving for 2026
This updated paper builds on Wakely’s prior analysis of Medicare Advantage benefit design, incorporating newly released 2026 enrollment data to provide a clearer picture of how market dynamics are evolving. With refreshed enrollment figures, the analysis reassesses which benefit strategies are gaining traction, where growth is concentrating, and how shifts in enrollment are aligning with plan design and competitive positioning.
LEAD: The Big Shift Beyond Attribution and Benchmarks-Quality as a Cash Flow Lever
The LEAD (the Long-term Enhanced ACO Design) Model is the Centers for Medicare & Medicaid Innovation Center’s newest accountable care organization (ACO)–focused model. Set to launch as ACO REACH (Realizing Equity, Access, and Community Health) sunsets at the end of 2026, LEAD directly links quality performance to benchmark dollars through a withhold, with 3% of the benchmark withheld upfront and only earned back through quality results. In practical terms, quality becomes a revenue protection lever. This paper identifies the changes in the LEAD compared to the ACO Reach model; ways the model is changing the financial conversations, and organizational readiness for these changes.
RFP Calendar
RFP Calendar
| Date | State/Program | Event | Beneficiaries |
|---|---|---|---|
| Date: February 2026 - DELAYED | State/Program: Illinois | Event: Awards | Beneficiaries: 2,400,000 |
| Date: March 20, 2026 | State/Program: Hawaii Community Care Services | Event: Proposals Due | Beneficiaries: 5,500 |
| Date: April 10, 2026 | State/Program: Hawaii Community Care Services | Event: Awards | Beneficiaries: 5,500 |
| Date: May 1, 2026 | State/Program: Nevada Children's Specialty | Event: Proposals Due | Beneficiaries: NA |
| Date: May 12, 2026 | State/Program: Nevada CO D-SNP | Event: Awards | Beneficiaries: 88,000 |
| Date: June 24, 2026 | State/Program: Wisconsin LTC GSR 3 | Event: Awards | Beneficiaries: 56,000 (all GSR) |
| Date: Summer 2026 | State/Program: Illinois Foster Care | Event: RFP Release | Beneficiaries: 33,000 |
| Date: July 1, 2026 | State/Program: Hawaii Community Care Services | Event: Implementation | Beneficiaries: 5,500 |
| Date: July 28, 2026 | State/Program: Nevada Children's Specialty | Event: Awards | Beneficiaries: NA |
| Date: August 2026 | State/Program: Indiana | Event: RFP Release | Beneficiaries: 1,400,000 |
| Date: January 1, 2027 | State/Program: Illinois | Event: Implementation | Beneficiaries: 2,400,000 |
| Date: January 1, 2027 | State/Program: Nevada CO D-SNP | Event: Implementation | Beneficiaries: 88,000 |
| Date: January 1, 2027 | State/Program: Wisconsin LTC GSR 3 | Event: Implementation | Beneficiaries: 56,000 (all GSR) |
| Date: January 1, 2027 | State/Program: Illinois Tailored Care Management Program | Event: Implementation | Beneficiaries: 22,400 |
| Date: July 1, 2027 | State/Program: Nevada Children's Specialty | Event: Implementation | Beneficiaries: NA |
| Date: January 1, 2028 | State/Program: Wisconsin LTC GSR 4,6 | Event: Implementation | Beneficiaries: 56,000 (all GSR) |
| Date: Fall 2027 | State/Program: Oregon | Event: RFP Release | Beneficiaries: 1,200,000 |
| Date: 2028 | State/Program: North Carolina | Event: RFP Release | Beneficiaries: 2,200,000 |
| Date: 2029 | State/Program: California | Event: RFP Release | Beneficiaries: NA |