As 2025 draws to a close, Congress finds itself at a crossroads on several critical health policy issues, with the fate of the Affordable Care Act (ACA) subsidies front and center. The year has been marked by intense negotiations and a flurry of proposals, many of which remain unresolved as lawmakers look ahead to a pivotal January 30 deadline for appropriations spending bills. In this article, policy experts from Health Management Associates (HMA)—including Leavitt Partners, an HMA company—provide a comprehensive wrap-up of Congress’ work on ACA subsidies, executive agency actions, and what stakeholders should anticipate in early 2026.
ACA Subsidies: A Year of Uncertainty and Political Maneuvering
The expiration of enhanced ACA subsidies at the end of 2025 has been a focal point for congressional debate. Despite numerous bipartisan groups and a multitude of proposals circulating, consensus has proven elusive. The Senate voted on an ACA-related measure December 11, 2025, but neither the Democrats’ proposal for a three-year extension nor the Republican alternative to replace subsidies with health savings accounts advanced and revise certain other Medicaid policies.
The situation in the House has been equally complex. House GOP leaders unveiled a healthcare package designed to lower costs, expand association health plans, and increase transparency for pharmacy benefit managers. The package would not extend the expiring enhanced ACA subsidies, and even if the House bill passes, the Senate is unlikely to consider it. In addition, on December 17, House Democrats secured enough support to force a vote on a bill that would provide a three-year extension of enhanced subsidies, although House rules preclude scheduling a vote on the bill until January.
The prevailing sentiment among policy experts is that no substantial action will be taken before year’s end.
The White House briefly floated a two-year extension of the enhanced subsidies, but walked back the proposal, signaling fluidity in the policy discussions within the administration and among congressional Republicans. The absence of consensus on both policy and political ramifications has left the ACA subsidy issue in limbo.
Looking Ahead: January’s Appropriations Deadline and ACA Options
December 15, 2025, marked the last day for consumers to enroll in ACA coverage policies that take effect January 1, 2026, meaning that for many health insurance purchasers, choices for 2026 are already set. Policymakers are now focused on another deadline for potential ACA subsidy action—January 30, 2026, when temporary funding for the current federal fiscal year expires. It is possible that a solution could be attached to the spending package, potentially affecting 2026 premiums, although operational challenges abound. The most feasible option at this stage would be a premium rebate, which would avoid reopening enrollment but require complex rate adjustments. Any substantive changes to the subsidy structure would demand significant actuarial analysis and could disrupt both health plans and state activities.
Congressional Dynamics: Appropriations, Extenders, and Policy Riders
The appropriations process is center stage as Congress approaches the January 30, 2026, deadline. Lawmakers are seeking to continue passing “minibus” packages—small groups of appropriations bills—to avoid another government shutdown. Most Medicare and Medicaid policy priorities, including must-pass extenders like telehealth flexibilities and the hospital at home program, are dependent on appropriations vehicles to advance. If Congress resorts to a stopgap continuing resolution, only the most essential extenders are likely to be included, with broader policy riders at risk of being sidelined.
Policy Outlook
Pharmacy benefit manager (PBM) reform stands out as a top bipartisan priority, with both House and Senate members eager to advance transparency and de-linking measures. Other lingering issues from the December 2024 healthcare package include Medicaid spread pricing prohibitions, streamlined enrollment for out-of-state providers, and targeted benefits for military service members. In Medicare, multi-cancer early detection screening and digital health policies may resurface, though larger reforms like Medicare physician fee schedule changes are likely to be deferred until later in 2026.
Agency Developments: CMS Innovation and Regulatory Changes
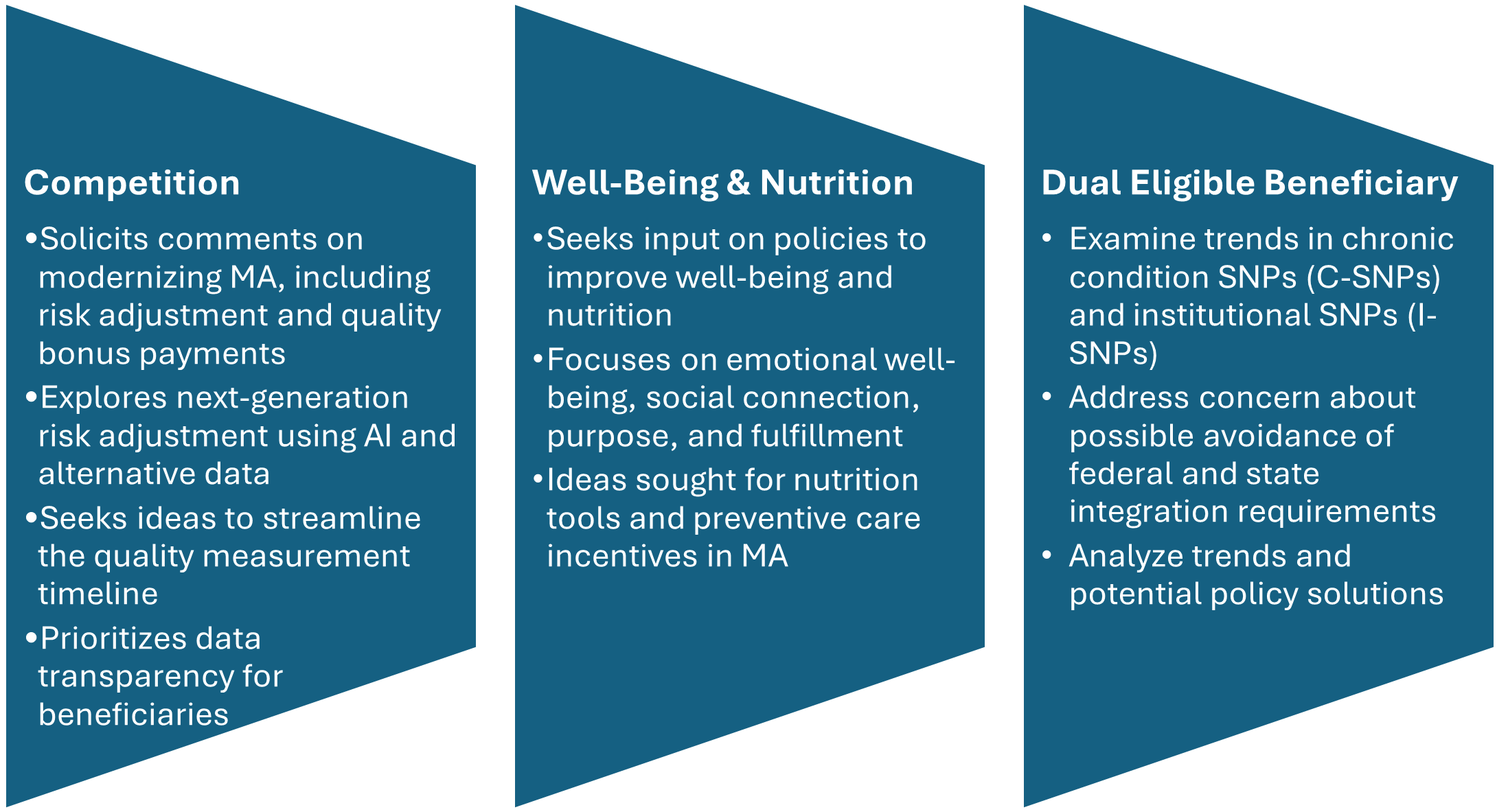
Beyond Congress, the Centers for Medicare & Medicaid Services (CMS) has been active, rolling out new models and rules that will shape the landscape in 2026 and beyond. Highlights include the 2027 Medicare Advantage Policy and Technical Changes Proposed Rule. Although it introduces no major policy shifts, the proposed rule addresses quality measurement, special needs plans, the Health Equity Index, and administrative burden reduction. It also codifies changes from the Inflation Reduction Act, such as cost-sharing and out-of-pocket limit reforms. The new ACCESS model (Advancing Chronic Care with Effective, Scalable Solutions) is intended to incentivize tech-enabled care for chronic conditions, with the model beginning July 2026.
CMS also released updates to the outpatient, home health, and durable medical equipment rules, with a continued focus on site neutrality (aligning payments across settings) and removing barriers to beneficiary choice. The agency is placing ongoing emphasis on data collection, price transparency, and updated payment methodologies to reflect modern practice and technology. The GENEROUS (GENErating cost Reductions fOr U.S. Medicaid) Model introduces most favored nation pricing for Medicaid, while additional mandatory Medicare drug pricing models are under review. Rural health transformation remains a CMS priority, with expectations for further announcements and awards before the end of the year.
We expect 2026 to be another busy year for CMS with more new models being announced, continued policy refinements in the fee-for-service payment systems, and changes in Medicare Advantage based on feedback from the requests for information.
Connect with HMA Policy Experts
As the new year approaches, uncertainty remains the defining feature of federal health policy. The fate of ACA subsidies, the appropriations process, and a host of other reforms will hinge on negotiations in the coming weeks. For stakeholders navigating these complex dynamics, HMA’s team of policy experts stands ready to provide guidance, analysis, and support.