Weekly Roundup -
January 14, 2026
Smart. Strategic. Essential.
Unmatched Healthcare Insights from HMA,
Leavitt Partners & Wakely.
Featured:
Webinar Replay – 2027 ACA Considerations: Proposed NBPP and Other Key Changes and Trends
ACCESS WEBINARTrending: In Focus
Tracking Medicaid’s Growth: FFY 2025 Spending and T-MSIS Data Provide Insights on Managed Care Spending
This week, our In Focus section highlights findings from a Health Management Associates Information Services (HMAIS) analysis of the Centers for Medicare & Medicaid Services (CMS) preliminary CMS-64 Medicaid expenditure report for federal fiscal year (FFY) 2025. The data show total medical services expenditures reached $971.4 billion across all states and territories, up 6.9 percent from FFY 2024.
This CMS-64 spending detail provides important context as states prepare for their upcoming legislative sessions and begin implementing changes required under the 2025 budget reconciliation act (P.L. 119-21, OBBBA). Early fiscal and operational pressures will stem from changes to the Supplemental Nutrition Assistance Program (SNAP) and preparations for community engagement requirements for Affordable Care Act (ACA) Medicaid expansion enrollees. In subsequent years, pressures will intensify because of major changes to provider tax financing and new federal limits on state directed payments in 2027 and early 2028.
In this article, we provide a deeper review of Medicaid spending, including the federal-state financing split. As Medicaid agencies prepare for upcoming spring sessions and anticipate potential program changes under OBBBA, it is notable that nearly two-thirds of Medicaid directors report an at least fifty percent likelihood of a Medicaid budget shortfall in FFY 2026.
Growth and Drivers in Medicaid Managed Care Spending
The HMAIS analysis looks at CMS-64 preliminary estimates of Medicaid spending by state for FFY 2025. CMS tracks state expenditures through the automated Medicaid Budget and Expenditure System/State Children’s Health Insurance Budget and Expenditure System (MBES/CBES).
While enrollment decreased for most states following the COVID-19 public health emergency unwinding, states saw an uptick in expenditures due to increased state directed payments, greater utilization and sicker populations, higher drug costs, increased provider rates, and greater use of long-term services and supports and behavioral health.
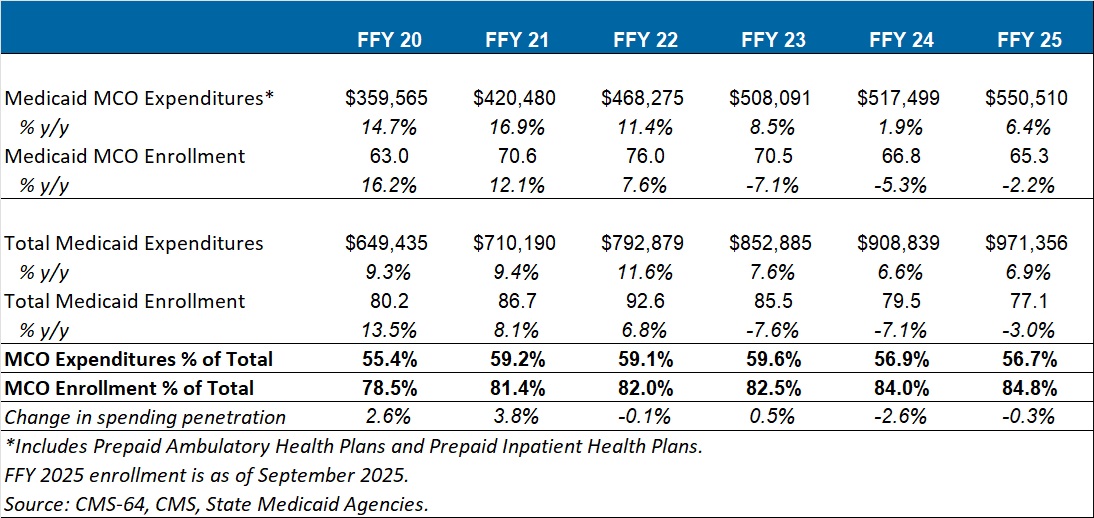
Key findings from HMAIS’ analysis (see Table 1), include:
- Total Medicaid managed care spending (federal and state share combined) reached $550.5 billion in FFY 2025, up from $517.5 billion in FFY 2024.
- This amount represents a 6.4 percent year-over-year increase from FFY 2024 to FFY 2025.
- Managed care accounted for 56.7 percent of total Medicaid spending in FFY 2025, down 0.3 percentage points from the previous year.
- The $33 billion increase from FFY 2024 to FFY 2025 exceeds the $9.4 billion increase seen the year prior, reflecting renewed growth following the unwinding transition period.
These figures include spending on comprehensive risk-based managed care organizations (MCOs), prepaid inpatient health plans (PIHPs), and prepaid ambulatory health plans (PAHPs). PIHPs and PAHPs refer to prepaid health plans that provide a subset of services, such as dental or behavioral health care. This total is exclusive of fee-based programs such as primary care case management models.
Table 1. Medicaid MCO Expenditures as a Percentage of Total Medicaid Expenditures, FFY 2020–2025 (in millions)
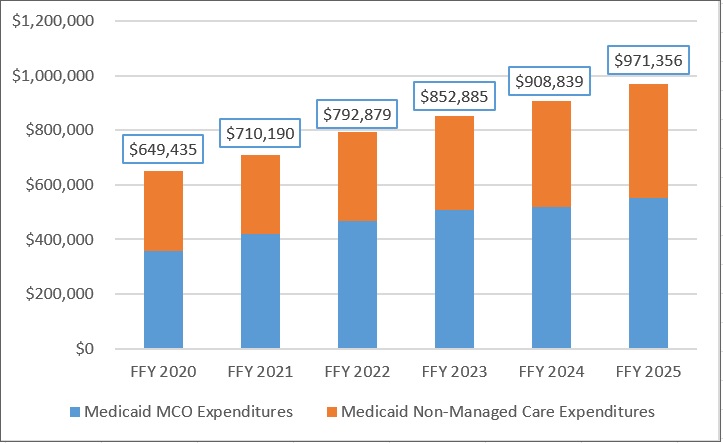
Annual Medicaid managed care expenditures have grown consistently with total Medicaid expenditures. After slower growth in FFY 2024—which aligned with the post-COVID-19 policy unwinding period when many states completed eligibility redeterminations—FFY 2025 again experienced an uptick in managed care growth (see Figure 1).
Figure 1. Total and MCO Medicaid Expenditures, FFY 2020–2025 ($M)
Federal versus State Share Spending
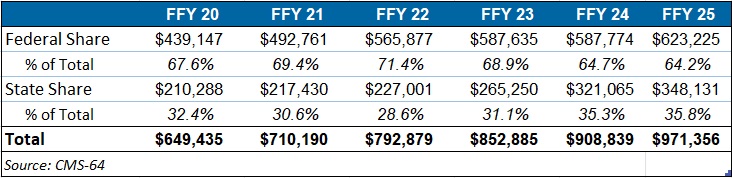
The preliminary FFY 2025 expenditure data provides a baseline before OBBBA’s changes are scheduled for implementation and as states continue to face Medicaid funding challenges. In FFY 2025, federal funding accounted for 64.2 percent of FFY 2025 spending, and non-federal matching funds accounted for 35.8 percent (see Table 2). Particularly later in 2027, 2028, and subsequent years, Medicaid expansion states stand to see disproportionally larger increases in their share of spending.
Table 2. Federal versus State Share of Medicaid Expenditures, FFY 2020–2025 (in millions)
T-MSIS Data Adds Detail to CMS-64 MCO Spending
To complement CMS-64 macro-spending trends, HMA developed a methodology allowing us to use Transformed Medicaid Statistical Information System (T-MSIS) data to approximate managed care spending by service category. Although T-MSIS enables more granular views (e.g., professional services, inpatient/outpatient hospital services, skilled nursing facilities (SNFs), HCBS, clinics, pharmaceuticals), the most recent dataset typically lags one to two years behind CMS-64 totals.
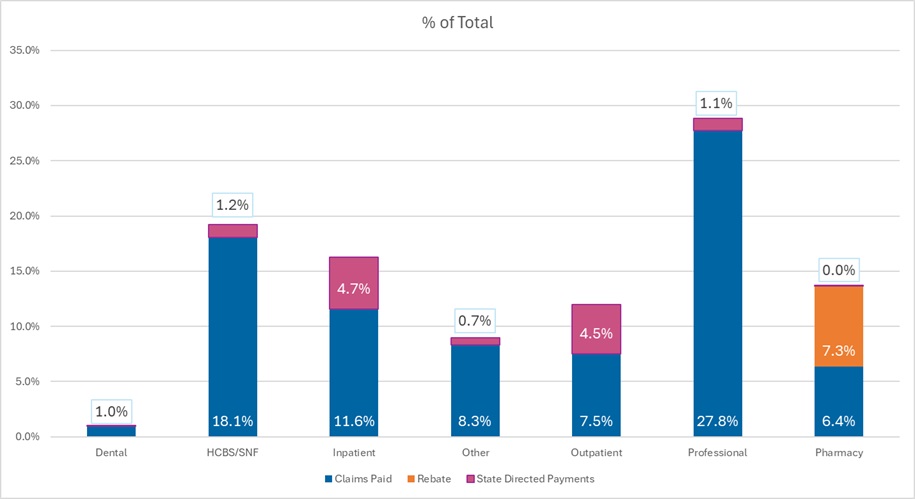
HMA’s analysis of the T-MSIS data shows that while managed care remains the dominant delivery system model for Medicaid, spending by provider types helps contextualize the CMS-64 report. Notably, the CMS-64 reports FFY25 data and our report below on T-MSIS disaggregation uses 2023 data. Although the T-MSIS and CMS-64 data are for different years, it still highlights the main components of the largest spending component of the CMS-64 with more recent data.
The 2023 T-MSIS analysis shows the following:
- Professional fees are the lead spending category, with nearly 30 percent of spending directed toward payments to physicians and other practitioners (e.g., physician assistants, nurse practitioners). Given that T-MSIS data are built around billing codes, services that traditionally may be considered part of a bundled rate (i.e., a large portion of physician services delivered in hospitals and clinics) are essentially unbundled and considered professional fees.
- Hospital spending (inpatient plus outpatient), SNF costs, and professional fees together account for close to 75 percent of spending in CY 2023.
Figure 2. T-MSIS Medicaid Spending by Service Category 2023 (MCO disaggregated plus FFS)
What to Watch
Because Medicaid is such a big part of state government spending, outlays for Medicaid will always be a focus and challenge for states. Upcoming state legislative sessions and OBBBA driven changes will begin in 2026 with SNAP pressures and major operational preparations for community engagement requirements for expansion states. Preparations for new limits on provider taxes and state directed payments will likely begin immediately, but the true impacts will occur in 2027 and early 2028. States will need to tailor their programs under funding constraints.
Connect with Us
HMAIS, a subscription-based tool that Health Management Associates offers, provides state-by-state analysis of the CMS-64 data, Medicaid managed care enrollment trends, and state budget reporting. For more information about an HMAIS subscription, contact Andrea Maresca and Alona Nenko. For details on T-MSIS data, contact Matt Powers and Shreyas Ramani.
Outlook 2026: What to Watch
As we kick off the new year, Health Management Associates (HMA) is launching a new series of brief, insightful interviews with our policy experts on issues that will define 2026—what’s changing, why it matters, and how federal, state, and industry decisions will shape what happens next. Building on our earlier analysis of the Rural Health Transformation Program ((RHTP), here and here), this week, we start with a pointed look at the Centers for Medicare & Medicaid Services’s (CMS) first year of RHTP awards.
Rural Health Transformation Program: What the Awards Signal for 2026
An interview with Kathleen Nolan, Senior Advisor, HMA, and Sara Singleton, Principal, Leavitt Partners, an HMA Company.
Q: What do the new Rural Health Transformation Program awards tell us about US Department of Health and Human Services (HHS) and CMS priorities heading into 2026?
Kathleen Nolan: One of the clearest signals is that CMS expects visible progress in 2026. This is not a program that gives states months of planning runway. The application made it clear that CMS wants states to start doing the activities they proposed right away—not just planning or propping up existing systems. CMS wants to see meaningful movement on implementation in 2026, especially in the areas of workforce, infrastructure, technology modernization, and care delivery redesign.
Sara Singleton: Exactly, and CMS is using this investment to reinforce some of the administration’s broader policy goals. Many state proposals leaned heavily into chronic disease prevention, chronic care management, and expanding supports that promote healthier lifestyles. That alignment isn’t accidental. The Administration is looking for real traction on these priorities, and RHTP gives states both the resources and the accountability framework to make progress. So, the message from CMS is clear: Move quickly, implement strategically, and show early gains in the areas that matter for long-term population health.
Q: Was anything in the awards themselves surprising?
Singleton: There was a lot of speculation about how wide the spread in funding levels might be, particularly for states’ discretionary initiatives. But the distribution was relatively tight; 32 states fell in the “average” range of $190‒$230 million, with only four states above $230 million and 13 below $190 million. That suggests CMS isn’t signaling dramatic differences in expected performance or ambition.
Nolan: It reinforces that CMS is looking for consistent, measurable progress from every state. States that struggle to implement their plans could see less funding in about years.
Q: What should states keep top of mind heading into year one?
Nolan: Accountability. CMS has made it clear they will adjust budgets in later years if states don’t meet expectations on reporting and evaluation. That also means states need to know where the dollars are going and what they are getting for the investment. Year one performance really matters.
Singleton: And it’s not just CMS. Congress and the Office of Inspector General for HHS will also be watching how states use these funds.
Q: What rural health policy developments are you watching in early 2026?
Nolan: Decisions about the leadership for these initiatives and state legislatures. Federal investment can only go so far. States will need strong leaders and supportive policies to accelerate and sustain RHTP efforts in year one. What legislatures choose to prioritize will shape the impact of RHTP far beyond year one.
Federal Policy News
Fueled By Leavitt Partners Weekly Health Intelligence
FDA Updates Guidance on Digital Health Technologies
On January 6, FDA released two final guidance documents related to digital health technologies, updating its policies on general wellness products and clinical decision support (CDS) software. The guidances supersede versions from 2019 and 2022, respectively, and are intended to provide updated clarity on products the agency intends to exclude from active regulatory oversight:
- In the General Wellness: Policy for Low Risk Devices guidance, CDRH clarified that it does not intend to examine low risk products intended solely for general wellness use, as defined in the guidance, and that present a low risk to the safety of users and other persons. Low risk products could include those for weight management, physical fitness, relaxation, or mental acuity. The guidance explicitly notes that products using non-invasive sensing to estimate physiologic parameters (g., blood pressure, oxygen saturation, blood glucose) may qualify as general wellness products if they are not intended for diagnosis or treatment and do not prompt specific clinical action. This update also addresses recent ambiguity regarding consumer wearables and permits the display of clinical-style metrics without premarket review if no specific diagnostic claims are made.
- The Clinical Decision Support Software guidance clarifies the scope of FDA oversight regarding CDS software intended to support, rather than replace, the judgement of health care professionals (HCPs). The guidance outlines the four criteria required for CDS software functions to be excluded from the definition of a device: (1) not intended to acquire, process, or analyze a medical image or a signal; (2) intended for the purpose of displaying, analyzing, or printing medical information; (3) intended to support or provide recommendations to an HCP about prevention, diagnosis, or treatment; and (4) intended to enable the HCP to independently review the basis for such recommendations so they do not rely primarily on the software. Notably, FDA stated it intends to exercise enforcement discretion for software functions that provide a single, clinically appropriate recommendation, provided the software meets all four exclusion criteria and enables the HCP to independently review the basis for the recommendation.
In a video posted to X, FDA Commissioner Makary discussed the new guidances, stating that they are intended to promote innovation and reduce regulatory barriers for non-medical grade products, for which FDA is looking to “get out of the way as a regulator.”
Trump Administration Cuts Billions in Mental Health, Addiction Grants
On January 14, 2026, the US Department of Health and Human Services cancelled billions of dollars in grant funding resources for substance use and mental health, effective immediately. The cancellations, issued by the Substance Abuse and Mental Health Services Administration (SAMHSA), affect front-line providers delivering overdose prevention, naloxone distribution, peer recovery, and crisis services, with organizations warning of immediate program closures.
ASPR Sets Five-Year Strategy for Preparedness and Response
On January 9, the Administration for Strategic Preparedness and Response (ASPR), released the agency’s Strategic Plan for 2026–2029. The ASPR Strategic Plan includes details on objectives across the five core goals, which are aligned with ASPR’s core beliefs, described as “anchor[ing] national health security by reinforcing partnership, gold standard science, stewardship, efficiency, accountability, and transparency.” The Strategic Plan continues to emphasize Trump Administration priorities, including state and local preparedness efficiency consistent with Executive Order 14239, reducing reliance on animal testing through the use of other research models, and emphasizing pharmaceutical onshoring. The Strategic Plan emphasizes five core goals for 2026–2029:
- Goal 1: Strengthen the Workforce and Reinforce a “One ASPR” Culture, under which ASPR states that it will seek to “promote a culture of information and resource sharing, collaboration, and transparency,” and eliminate siloes across its various centers;
- Goal 2: Strengthen Preparedness through State and Local Resiliency, under which ASPR emphasizes its role in supporting the readiness of state, local, tribal, and territorial (SLTT) partners through guidance and programs, including the Hospital Preparedness Program (HPP) and National Special Pathogen System (NSPS);
- Goal 3: Execute Rapid, Efficient, and Adaptive Federal Response, under which ASPR seeks to strengthen several aspects of federal response, including the Strategic National Stockpile and federal medical response teams;
- Goal 4: Secure America’s Medical Supply Chain, under which ASPR notes efforts to onshore manufacturing of medical countermeasures, medical supplies, active pharmaceutical ingredients (APIs) and key starting materials (KSMs); and
- Goal 5: Address Emerging Health Security Threats, in which ASPR states several new objectives including leveraging digital health and AI to improve threat assessment and enhance patient care.
Stakeholders engaging with ASPR should consider how policies and priorities fit within ASPR’s strategic plan. Additional details on how ASPR plans to execute its strategic plan may be included in future Congressional Budget Justifications.
Federal Dietary Guidelines Shift Focus to Food Quality
On January 7, USDA and HHS released the updated Dietary Guidelines for Americans, 2025–2030. The updated guidelines urge Americans to prioritize whole foods, such as fruits, vegetables, whole grains, nuts, seeds, and lean proteins, while reducing reliance on ultra-processed products. The agencies cite scientific evidence linking ultra-processed foods to obesity, diabetes, and heart disease, and indicated that future dietary guidelines will focus on food quality and dietary patterns. In the press release accompanying the updated guidelines, HHS and USDA criticize previous guidelines for failing to “take a hard line” regarding certain recommendations, such as added sugar consumption and ultra-processed foods, and for previous efforts to prioritize health equity. Alongside the updated guidelines, the Administration announced a new food pyramid and the site realfood.gov. Key changes resulting from updated dietary guidelines include procurement policies for federal facilities, school meal programs, and nutrition assistance programs. USDA and HHS state that they will work with farmers, food producers, schools, and local governments to support the transition and strengthen supply chains. The agencies also committed to ongoing evaluation, public input, and expanded investment in nutrition research and education.
FDA Expands CMC Flexibilities for Cell and Gene Therapies
On January 11, FDA provided information regarding the agency’s flexible approach to overseeing chemistry, manufacturing, and control (CMC) requirements for cell and gene therapies (CGT). CBER cites the innovative nature of CGT products and the unmet medical needs they often seek to address in FDA’s decision to provide additional regulatory flexibilities and seeks to clarify those through this announcement. The agency notes several flexibilities that will be permitted across three areas for CGT.
- Clinical Development Flexibilities, such as a sponsor will not be expected to comply with 21 CFR part 211 before an investigational product is manufactured for phase 2 or 3 clinical trials, and as sponsors move from phase 1 to studies designed to establish efficacy for licensure, minor manufacturing changes supported by data showing the comparability of the pre-change and post-change product may be permitted.
- Commercial Specifications Flexibilities, such as flexibility will be considered in establishing product release specifications in the review of CGT BLAs given a small patient populations targeted by CGT therapies do not necessarily allow for a large number of manufacturing lots.
- Process Validation Flexibilities, such as permitting specific Process Performance Qualification (PPQ) lots to be designed by batch for release and distribution before protocol execution steps are completed, and flexibility in consideration for whether PPQ protocols justify the appropriate number of lots based on overall process understanding.
In a video discussing the announcement, Commissioner Makary specifically noted that the agency will no longer require a sponsor to supply three Process Performance Qualification (PPQ) lots for process validation, which FDA has assessed as a particular “pain point” for companies.
Ready to talk about your organization's challenges?
Schedule a ConsultationState Policy News
California
California Ends Medicaid Coverage of Weight Loss Drugs. KFF Health News reported on January 9, 2026, that California has ended Medicaid coverage of GLP-1 weight loss drugs, effective January 1, 2026. The state had projected GLP-1 costs to quadruple over four years to about $800 million per year if it had not terminated coverage. The state will still cover GLP-1s for other health issues.
Kentucky
Kentucky Medicaid Oversight Board Recommends Transparency Reforms. WLKY reported on January 12, 2026, that Kentucky’s bipartisan Medicaid Oversight and Advisory Board concluded that Medicaid spending has risen significantly without corresponding improvements in health outcomes and that transparency and performance reporting are fragmented. The board recommended creating a centralized public dashboard and strengthening performance standards and data access to improve oversight, accountability, and readiness for upcoming federal policy changes.
Minnesota
Minnesota Appeals CMS Withholding of $2 Billion in Medicaid Funds. The Minnesota Department of Human Services announced on January 13, 2026, that it is appealing a Trump administration decision to withhold more than $2 billion a year in Medicaid funding, announced by Centers for Medicare & Medicaid Services (CMS) Administrator Dr. Mehmet Oz, who cited alleged shortcomings in the state’s efforts to combat Medicaid fraud. State officials say the allegations are unfounded and that Minnesota has already taken extensive program integrity actions, including auditing high-risk providers, suspending certain services, imposing provider moratoria, and strengthening payment reviews. The state argues the funding freeze violates federal rules, could destabilize its healthcare system, and would affect 1.2 million Medicaid enrollees, and has formally requested a hearing to challenge the decision.
New York
New York Governor Proposes Safety Net Funding, Behavioral Health Investments in State of the State Address. New York Governor Kathy Hochul released on January 13, 2026, the 2026 State of the State Address, where she outlined her priorities for the next year. Her proposals include providing additional funding through the safety net transformation program, creating more transparency for prior authorizations, and strengthening oversight of the Material Transactions Law. Hochul will direct the state Department of Health (DOH) to identify high-cost drug classes for direct price negotiations with manufacturers and to make the Certificate of Need (CON) process more efficient. The governor wants to invest in behavioral health through a three-year suicide prevention plan, increased funding for supportive housing programs, teen mental health first aid training, and other youth behavioral health investments. Additionally, Hochul proposes improving access to end of life care, launching the Community Aging in Place – Advancing Better Living for Elders (CAPABLE) program, and investing $250 million for new affordable housing.
West Virginia
West Virginia Finance Committee Hears Conflicting Reports on Moving LTSS to Managed Care. The Weirton Daily Times reported on January 13, 2026, that West Virginia legislators on the Joint Standing Committee on Finance heard from the executive director of the West Virginia Association of Health Plans, which represents the state’s four Medicaid managed care organizations, on ways to reduce healthcare costs for Medicaid services. Some of the key recommendations include integrating long-term services and supports (LTSS) into managed care, expanding telehealth access, standardizing provider fee schedules, reviewing the Certified Community Behavioral Health Clinic program, and reviewing pharmacy benefits. However, the West Virginia Health Care Association pushed back on the report, stating that shifting LTSS to managed care would “create unnecessary bureaucratic red tape without improving health outcomes.” The association argued the expansion of managed care organizations within the state has coincided with escalating costs and worsening health rankings.
Private Market News
Fueled By Wakely Consulting Group
ACA Marketplace Enrollment Dips 3.5 Percent Year-Over-Year
On January 12, 2026, the Centers for Medicare & Medicaid Services (CMS) published preliminary data showing Affordable Care Act (ACA) Marketplace enrollment is down approximately 3.5 percent from a year ago, with roughly 22.8 million people enrolled for 2026 to date.
On the Hook for Uninsured Residents, Counties Now Wonder How They Will Pay
On January 7, Fierce Healthcare reported that county health officials across the country are preparing for an estimated ten million people to become uninsured over the next decade as federal funding cuts and policy changes roll back coverage expansion from the Affordable Care Act leaving local governments uncertain how they will finance care for residents without insurance.
Our Insights
Fueled By Experts Across Our HMA Companies
Health Management Associates
Webinar: 2027 ACA Considerations: Proposed NBPP and Other Key Changes and Trends
Upon the release of the CMS final 2027 Notice of Benefit and Payment Parameters and the accompanying Letter to Issuers in January, health plans and state policymakers will face critical decisions that shape the next phase of the individual and small group markets. Join experts from HMA and Wakely for a timely discussion unpacking what the proposed rule means in practice and how stakeholders can begin preparing now.
Webinar: The ACCESS Model: Essentials for Applicants
CMS’s new ACCESS model represents one of the most ambitious federal efforts to modernize chronic care through technology-supported services. This national, voluntary, decade-long model creates a new payment pathway for digital health tools, continuous monitoring, behavioral support, and other tech-enabled interventions that complement traditional care. With beneficiaries able to enroll directly and clinicians eligible for co-management payments, ACCESS introduces a fundamentally different approach to chronic condition management across Medicare. In this webinar, HMA and Leavitt Partners experts will break down what is known today, what to expect in the forthcoming Request for Applications, and what organizations can do to prepare. initiative.
Webinar: Meeting the Healthcare Needs of Unhoused People Part 1: Service and Care Responses
Join HMA experts and our featured speakers for the first of two webinars exploring how current events are impacting people experiencing homelessness and their access to care. This webinar will highlight the model of care for healthcare for the homeless clinics and medical respite care providers and how these services interact with broader systems of care. Additionally, we will explore how the current environment is impacting delivery and financing of care for some of our most vulnerable neighbors.
Webinar: Meeting the Healthcare Needs of Unhoused People Part 2: State Policy Responses
Recent federal policy changes, such as the 2025 Budget Reconciliation Act (OBBBA), bring significant challenges to retaining the Medicaid coverage gains and added 1115 demonstration services that have been so successful in the last decade. States will be under tremendous pressure to meet new requirements—but they also have options to reduce the negative impact on vulnerable populations and the healthcare providers that serve them. Join HMA and our featured experts for this webinar to discuss state-level policy options, share resources, and consider how to move forward in the current environment.
Wakely
When Stars Realign: Understanding CMS’s 2027 Proposed Rule
This white paper provides an overview and impact analysis of the Star Rating changes proposed in the 2027 Medicare Advantage and Part D Proposed Rule issued by CMS. Findings include:
- Key components of the proposed Star Rating changes
- Analysis of impacts across major parent organizations and the market as a whole
- Brief discussion of the future of Star Ratings and how plans can prepare for upcoming changes
RFP Calendar
RFP Calendar
| Date | State/Program | Event | Beneficiaries |
|---|---|---|---|
| Date: DELAYED | State/Program: Texas STAR & CHIP | Event: Implementation | Beneficiaries: 4,600,000 |
| Date: December 2025 - February 2026 | State/Program: Texas STAR Kids | Event: Awards | Beneficiaries: 150,000 |
| Date: January 1, 2026 | State/Program: Wisconsin LTC GSR 2,7 | Event: Implementation | Beneficiaries: 56,000 (all GSR) |
| Date: January 1, 2026 | State/Program: Michigan HIDE SNP | Event: Implementation | Beneficiaries: 35,000 |
| Date: January 1, 2026 | State/Program: Nevada D-SNP | Event: Implementation | Beneficiaries: 88,000 |
| Date: January 1, 2026 | State/Program: Ohio Duals | Event: Implementation | Beneficiaries: 250,000 |
| Date: January 1, 2026 | State/Program: Illinois D-SNP | Event: Implementation | Beneficiaries: 79,000 |
| Date: January 1, 2026 | State/Program: Nevada | Event: Implementation | Beneficiaries: 674,000 |
| Date: January 1, 2026 | State/Program: Massachusetts One Care, Senior Care Options | Event: Implementation | Beneficiaries: 120,000 |
| Date: January 6, 2026 | State/Program: Nevada Children's Specialty | Event: Proposals Due | Beneficiaries: NA |
| Date: January 16, 2026 | State/Program: Wisconsin LTC GSR 3 | Event: Proposals Due | Beneficiaries: 56,000 (all GSR) |
| Date: January 21, 2026 | State/Program: Illinois Tailored Care Management Program | Event: Proposals Due | Beneficiaries: 22,400 |
| Date: February 2026 | State/Program: Illinois | Event: Awards | Beneficiaries: 2,400,000 |
| Date: February 19, 2026 | State/Program: Nevada Children's Specialty | Event: Awards | Beneficiaries: NA |
| Date: June 24, 2026 | State/Program: Wisconsin LTC GSR 3 | Event: Awards | Beneficiaries: 56,000 (all GSR) |
| Date: December 2026 - February 2027 | State/Program: Texas STAR Kids | Event: Implementation | Beneficiaries: 150,000 |
| Date: January 1, 2027 | State/Program: Illinois | Event: Implementation | Beneficiaries: 2,400,000 |
| Date: January 1, 2027 | State/Program: Nevada Children's Specialty | Event: Implementation | Beneficiaries: NA |
| Date: January 1, 2027 | State/Program: Wisconsin LTC GSR 3 | Event: Implementation | Beneficiaries: 56,000 (all GSR) |
| Date: January 1, 2027 | State/Program: Illinois Tailored Care Management Program | Event: Implementation | Beneficiaries: 22,400 |
| Date: January 1, 2028 | State/Program: Wisconsin LTC GSR 4,6 | Event: Implementation | Beneficiaries: 56,000 (all GSR) |
| Date: Fall 2027 | State/Program: Oregon | Event: RFP Release | Beneficiaries: 1,200,000 |
| Date: 2028 | State/Program: North Carolina | Event: RFP Release | Beneficiaries: 2,200,000 |