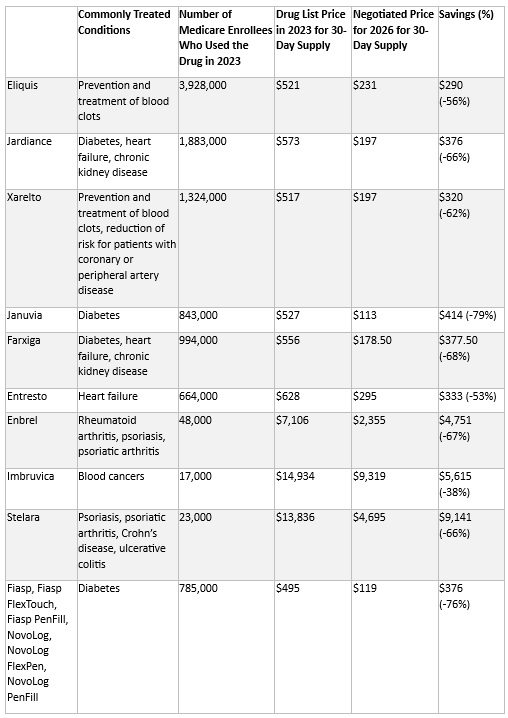
Our second In Focus article reviews the recently proposed Notice of Benefit and Payment Parameters (NBPP) for 2026. The Centers for Medicare & Medicaid Services (CMS) proposed rule, released October 10, 2024, describes the policy and payment changes that will affect the Affordable Care Act (ACA) markets in 2026. Public comments must be submitted to CMS by November 12, 2024. Key highlights from the proposed rule follow.
Broker Oversight and Monitoring
CMS proposes to increase oversight and accountability for brokers and agents that write policies through HealthCare.gov. In response to the discovery earlier this year of fraudulent actors reassigning broker designations and switching consumer enrollments without their permission or knowledge, CMS has already implemented several corrective actions, including the suspension of 850 Healthcare.gov agents and brokers. CMS intends to build on these actions through the following interventions:
- Clarify that lead agents, typically an agency owner or executive, are subject to the same rules as individual brokers, agents, and web-brokers and that enforcement action can be taken against the lead agents if they explicitly or implicitly condone misconduct or fraud
- Broaden CMS’s authority to suspend broker and agent system access, inclusive of instances of suspected misconduct that affects eligibility determinations, operations, applicants, or systems
- Update the model consent form to include documentation of the broker reviewing and confirming the accuracy of submitted application information with the consumer.
Marketplace User Fees
CMS proposes to increase the user fee collected to pay for administration of HealthCare.gov as follows:
- Between 1.8 percent and 2.5 percent in 2026 for federally facilitated marketplaces (FFM) states, up from 1.5 percent of monthly premiums in 2025
- Between 1.4 percent and 2 percent in 2026 for state-based marketplaces on the federal platform (SBM-FPs), up from 1.2 percent in 2025
The proposed changes are due, in part, to uncertainty caused by the future of the enhanced premium tax credits that are set to expire at the end of 2025. The enhanced premium tax credits are the driving force behind the increase in nationwide marketplace enrollment to more than 21 million people in 2020 from 11.4 million in 2020. If not extended, or if it takes past March 2025 for Congress to act, CMS has indicated the user fees will increase in 2026 to 2.5 percent for FFM states and 2% for SBM-FPs to accommodate expected enrollment declines. Notably, after several years of significant decreases, CMS is proposing to increase the user fees above 2025 levels regardless of the outcome of the enhanced premium tax credits.
Plan Limits for Non-Standard Plans
CMS proposes to clarify rules limiting the number of non-standardized plans an issuer can offer through HealthCare.gov (two or less in 2025). The limit is applied per product network type (e.g., HMO, PPO), per metal level, per service area, per inclusion of adult/pediatric dental and/or vision benefits (with additional exceptions, starting in payment year (PY) 2025, for plans with specific design features that would substantially benefit consumers and meet other requirements). To maximize the number of non-standardized plans offered on HealthCare.gov, an issuer could offer up to 16 plans per metal level and network type in a given service area by creating every combination of adult dental, pediatric dental, and adult vision (or even more, if plans meet the exception requirements).
Though CMS does not limit the number of standardized plan options an issuer offers on HealthCare.gov, they propose reinstating a meaningful difference standard to prevent consumer confusion and unnecessary plan proliferation. The proposed standard is similar to the removed standard from 2019; for plans in the same metal level, product type, and service area, a reasonable consumer needs to be able to identify at least one material difference in benefit coverage, provider networks, and/or formulary.
New Premium Payment Threshold Options for Issuers
CMS proposes new options for issuers to avoid triggering late payment grace periods for consumers who make most but not all of their premium payment to minimize termination of coverage for consumers who owe a small amount. The options include:
- The current option of a “reasonable” percentage of net premium. CMS proposes codifying 95 percent as the minimum threshold.
- New proposals of as low as 99 percent of gross premium and a fixed-dollar threshold of $5 or less.
CMS is also considering limiting issuers to offering just one payment threshold option—either fixed-dollar or percent of premium—to avoid consumer confusion.
Increased Transparency for State-Based Marketplaces
CMS proposes new initiatives to promote transparency into state-based marketplace (SBM) program operations. These initiatives include:
- Publishing State Marketplace Annual Report Tool (SMART) submissions, which are used to monitor SBM compliance with select eligibility and enrollment, program integrity, and financial reporting requirements. SBMs must annually participate in independent programmatic and financial audits as part of SMART. CMS proposes to make the 2023 SMART submissions public in spring 2025.
- Expanding the disclosure of SBM information to include data collected but not currently published, including details on SBM eligibility, enrollment, and plan certification policies as well as Navigator program spending, call center metrics, and website traffic data.
SBMs already are required to publish programmatic and financial audit summaries and generally publish robust data and information on their program operations through public reports and meetings; however, this information is neither centrally located nor consistently published across all SBMs.
Key Considerations
The proposed 2026 NBPP would build on previous actions that CMS has taken to address fraudulent broker and agent activity and to shore up financial sustainability of Healthcare.gov operations in light of uncertainty about the enhanced premium tax credits. It also seeks to make clear how plan variations adding dental or vision benefits factor into HealthCare.gov plan limits and gives issuers new premium payment threshold options. Lastly, it proposes new transparency requirements for SBMs. Interested stakeholders, including SBMs and issuers, should monitor how these proposed changes will affect consumers, operational processes, product strategy, and financial sustainability.
Connect With Us
The Health Management Associates, Inc., team has the depth, experience, and subject matter expertise to assist with tailored analysis and the modeling capabilities to assess the policy impacts to consumers, marketplaces, and issuers. If you have questions or want to discuss the proposed rule, contact our featured experts below.
For additional information on elements of the proposed NBPP not discussed here, Wakely Consulting’s white paper, Summary of Provisions of HHS’ Proposed 2026 Notice of Benefit and Payment Parameters and Other Key Regulations, highlights the proposed changes to the Risk Adjustment program, Medical Loss Ratio, and the Actuarial Value Calculator, among other changes.