On January 28, 2026, the Centers for Medicaid & Medicare Services (CMS) released its second national snapshot of 2026 Affordable Care Act (ACA) Marketplace Open Enrollment (OE) activity. While this update is not a final accounting of enrollment activity, it is likely to be the last OE federal data release for some time and offers an early look at how enrollment trends are shifting in the wake of expired enhanced premium tax credits and new eligibility standards under the 2025 budget reconciliation act (P.L. 119-21, OBBBA).
In this article, Health Management Associates (HMA) and Wakely, an HMA company, highlight findings from their analysis of the 2026 OE activity and compare this activity with 2025 data. This analysis builds on the findings in their January 2026 analysis (here) and will provide important context for the 2027 plan year.
Overall Enrollment Trends
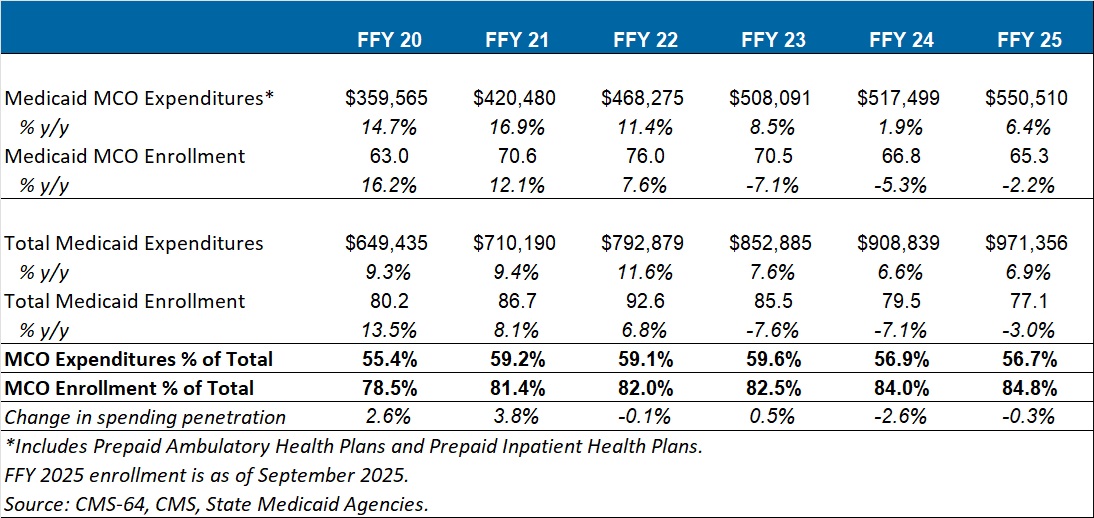
CMS reports that 2026 plan selections decreased by 5 percent from 2025, with enrollment declining across both new and returning consumers. New sign-ups dropped by 14 percent and renewals fell by 3 percent (Table 1). State-based Marketplace (SBM) enrollment dipped modestly, though many SBMs were still enrolling consumers in late January.
Table 1. Comparison of 2026 and 2025 Open Enrollment
| 2026 | 2025 | Net Change | |
| Total | 22,973,219 | 24,166,491 | (1,193,272) |
| New Consumers | 3,382,189 | 3,938,907 | (556,718) |
| Returning Consumers | 19,591,030 | 20,227,584 | (636,554) |
Variation Across State-Based and Federally Facilitated Marketplaces
Enrollment patterns varied substantially across states.
SBMs:
- New Mexico saw the largest year-over-year increase (14%), attributed to state-funded subsidies designed to offset the loss of enhanced premium tax credits (ePTCs).
- Georgia experienced a 14 percent decline, the steepest drop among SBMs.
Federally Facilitated Marketplace (FFM) States:
- Overall, FFM enrollment fell 5 percent.
- Texas led FFM states with a 5 percent increase in plan selections.
- Ohio and North Carolina experienced substantial enrollment declines, 20 percent and 22 percent respectively.
What This Tells Us—and What It Doesn’t Tell Us Yet
FFM data are as of January 15, 2026, and measure plan selections after the OE period ended. Within the FFM, state-by-state enrollment activity varied significantly. Some of this variation is surprising and not readily explainable from the available data and will be a focus of future HMA and Wakely analyses.
The data include neither effectuated enrollment nor paid enrollment—data which will be key to fully understanding 2026 enrollment trends and the impact of changing federal policies, including the ePTC expiration and changing eligibility standards introduced in 2026 as the result of OBBBA.
Early SBM data suggest significantly higher cancellation and disenrollment rates than in previous years.
SBMs are sharing that they expect substantial affordability-driven voluntary and nonpayment terminations over the first half of 2026.
Monitoring paid enrollments, attrition, and grace period dynamics, including retro-terminations, will be key to understanding market dynamics and 2027 pricing.
Connect with Us
HMA and Wakley experts have considerable experience working with states, insurers, and federal policymakers with jurisdiction over the Marketplace. We work with these entities to inform, analyze, and shape federal policies and conduct impact analyses on pricing, enrollment, administration, and operations. HMA also provides strategic and project management support for the implementation of finalized policies.
Please contact Michael Cohen, Taylor Gehrke, or Zachary Sherman with questions, follow-up, or if you would like expert assistance exploring any of the issues discussed in this post.