
March 4, 2026
The Value Shift in Medicare Advantage: What 2026 Benefits Tell Us About the Market’s Next Chapter
HMA Insights – including our new podcast – puts the vast depth of HMA’s expertise at your fingertips, helping you stay informed about the latest healthcare trends and topics. Below, you can easily search based on your topic of interest to find useful information from our podcast, blogs, webinars, case studies, reports and more.

The Value Shift in Medicare Advantage: What 2026 Benefits Tell Us About the Market’s Next Chapter

The Centers for Medicare & Medicaid Services (CMS) 2027 Notice of Benefit and Payment Parameters (NBPP) proposed rule, published February 11, 2026, arrived at a pivotal moment for the Affordable Care Act (ACA) Marketplaces. The temporary enhanced premium tax credits (ePTCs), first expanded in 2021 and extended through 2025, expired at the end of last year, returning Marketplace subsidies to their original ACA structure in 2026. As we discussed in earlier articles (here and here), that shift is already affecting affordability, plan selection, and enrollment dynamics—particularly for consumers who are ineligible for premium assistance.
The proposed 2027 NBPP represents a significant reset for the Marketplace, reflecting CMS vision and policy priorities to strengthen program integrity while expanding plan design flexibility and consumer choice as a pathway to affordability, as well as policies to defer to state authority. Healthcare organizations and other interested stakeholders may submit comments on the proposed rule through March 13, 2026.
The remainder of this article addresses the key policy proposals and considerations for issuers, states, and consumer groups.
CMS’s Proposals
The proposed NBPP for 2027 sets standards for the Exchanges and ACA-compliant individual and small group markets and updates payment parameters for risk adjustment and risk adjustment data validation (RADV). The rule also implements changes approved under the 2025 Budget Reconciliation Act, (P.L. 119-21, OBBBA) and includes a range of policies spanning plan certification, eligibility and verification, and Exchange oversight.
Expanded Plan Design Flexibility
CMS proposes to discontinue standardized plan options in the Federally-facilitated Marketplace (FFM) and remove limits on the number of non-standardized plans offered by issuers on the FFM and state-based Marketplaces on the federal platform (SBE-FPs). Issuers would be permitted to decide whether to discontinue existing standardized or chronic condition plans or continue them with modified cost sharing.
Considerations: This change is designed to allow greater innovation in plan design. It also raises questions about the potential return of a more complex Marketplace shopping experience for consumers who will have to shift through more plans.
Certification of Non-Network QHPs
One of the most consequential proposals would allow “non-network” plans to be certified as qualified health plans beginning in 2027. These plans would not rely on contracted provider networks. Instead, they would set benefit payment amounts and require issuers to demonstrate that sufficient providers—including Essential Community Providers (ECPs) and mental health and substance use disorder providers—are willing to accept those amounts as payment in full.
Considerations: CMS positions non-network plans as a way to create lower premium options. For states and issuers, this proposal introduces new oversight and operational considerations related to access standards, consumer protections, the risk of balance billing or access gaps for consumers, and potential market instability.
Changes in Catastrophic and Bronze Cost Sharing
The proposed rule would further expand access to catastrophic plans by codifying hardship exemptions for individuals ineligible for advance premium tax credits (APTCs) or cost-sharing reductions (CSRs) because of projected income. CMS also proposes to allow multiyear catastrophic plans with contract terms of up to 10 consecutive years. In addition, CMS proposes new flexibility for certain bronze plan designs in the individual market. In both cases, CMS proposes to allow catastrophic and bronze plans to exceed the annual maximum out-of-pocket limit.
Consideration: These policies reflect CMS’s emphasis on affordability through lower premiums and expanded consumer choice, while shifting more financial risk to enrollees through higher cost sharing.
Network Adequacy and Essential Community Providers
CMS proposes to give states greater discretion in provider access for network adequacy and ECP certification reviews, including allowing federally funded exchange (FFE) states to conduct their own reviews if CMS determines they have sufficient authority and technical capacity. CMS also proposes to reduce the minimum percentage of ECPs that issuers must include in their networks from 35 percent to 20 percent.
Considerations: These changes reduce federal prescriptiveness and could lower issuer compliance costs but also place more responsibility on states to monitor access and ensure that vulnerable populations are not adversely affected.
Essential Health Benefits and State Mandates
The proposed rule would prohibit issuers from including routine non-pediatric (adult) dental services as an Essential Health Benefit (EHB). More significantly for states, CMS proposes changes to cost defrayal requirements for state-mandated benefits, requiring states to cover the cost of benefits considered “in addition to EHB” under specified criteria, even if those benefits are embedded in the state’s EHB benchmark plan.
Consideration: These changes could have direct budgetary implications for states, pricing implications for issuers, and could stunt or potentially decrease benefits for consumers.
Program Integrity and Increased Eligibility Verification
CMS includes a robust set of program integrity provisions, including:
Consideration: These policies continue CMS’s heightened scrutiny of enrollment activity and subsidy eligibility. CMS’s policies are likely to increase data matching issues (DMIs), which could increase burden on Marketplaces and enrollees, resulting in reduced enrollment.
Preparing for Policy Driven Changes in ACA Marketplaces
The 2027 NBPP underscores a clear policy shift away from extending federal subsidies toward advancing a Marketplace framework that emphasizes program integrity, state flexibility, and expanded plan design options as mechanisms to promote affordability and consumer choice.
The proposed rule sets the stage for significant strategic and operational decisions for issuers and states ahead of the 2027 plan year. Health Management Associates (HMA), including Wakely, an HMA company, works with issuers modeling enrollment and risk shifts and to assist in pricing decisions. States also should consider the need for new strategies and approaches to adapt to federal policy changes that are expected for ACA Marketplace programs.
For more information about the policies described in this article, support with scenario-based modeling of enrollment and data-informed strategy development for 2027 and beyond, please contact our experts Michael Cohen, Lina Rashid, or Zach Sherman.

In this conversation, Andrea Maresca, Senior Principal at Health Management Associates (HMA), caught up with Tim Courtney, Director, Wakely, and Jonathan Blum, Co-Founder & Managing Partner at Health Transformation Strategies, LLC, to unpack the biggest questions emerging from the Calendar Year (CY) 2027 Medicare Advantage (MA) and Part D Advance Notice. Of particular interest was the Centers for Medicare & Medicaid Services’s (CMS’s) proposed risk adjustment and diagnosis source changes, which are drawing significant attention across the industry.
Q: The headline is “flat” payments. How should the market interpret CMS’s projected rate change?
Tim Courtney: CMS projects a net average payment change of just +0.09 percent for CY 2027—about $700 million (M). The effective growth rate is about 4.97 percent, but it’s largely offset by risk model and normalization changes and the proposed diagnosis source policy.
Jon Blum: Exactly. It’s important to note that CMS’s impact projections are based on the change in its average payments. Its proposed policies will have much more far-reaching distributional impacts, depending on the diagnoses of their enrolled members. At the same time CMS recently proposed changes to its Star Ratings methodologies. Over time, we could see quite significant changes to the balance of Medicare Advantage payments distributed across the country that could significantly affect benefit offerings and premium amounts.
Q: What’s most surprising in the Advance Notice for 2027?
Blum: The diagnosis source tightening is the big one. CMS proposes excluding diagnoses from “unlinked Chart Review Records” from risk score calculation starting in CY 2027. That signals a continued progression by the agency toward encounter-anchored data integrity. Assuming this policy is finalized, Medicare Advantage plans must continue to invest in systems to respond to CMS’s program integrity focus.
Courtney: And it’s not only chart review. CMS also proposes excluding diagnoses from audio-only services for Part C and similarly for Part D. Operationally, that’s a big deal. Plans need to understand where diagnoses originate, how they’re supported, and what the downstream risk adjustment factor (RAF) impact looks like by segment and provider channel.
Q: The Wakely team estimates a different “feel” than CMS’s topline. What does Wakely’s analysis add?
Courtney: Wakely’s summary helps translate CMS components into both benchmark and plan payment change.
Blum: This point is really key. Wakely’s analysis flags that rebasing/repricing impacts aren’t fully reflected yet, which means county-level outcomes can diverge materially once the final Rate Announcement is released. The rebasing could be particularly volatile this year as CMS adjusts for rural emergency hospital payments and the removal of anomalous and suspect DME claims. Both adjustments vary by geographic area.
Q: How should plans think about bid strategy and benefit pressure for 2027?
Courtney: The tighter risk adjustment environment could squeeze rebates and supplemental benefit richness—especially if bids don’t adjust quickly. Wakely estimates risk-adjusted bid and rebate revenue is down roughly 0.35 percent under a set of simplifying assumptions, underscoring the margin sensitivity.
Practically this means plans should run a few scenarios: 1) RAF compression from diagnosis source changes, 2) normalization updates, and 3) Star-related shifts—even if the Star change is estimated to be small nationally.
Blum: I’d add provider contracting and clinical program return on investment (ROI) will likely be an even greater focus for Medicare Advantage plans. When risk score lift is constrained, the value of medical cost management and quality performance becomes more important. We have seen tremendous pushback by healthcare providers over the greater use of prior authorization, with some major health systems dropping their contracts with Medicare Advantage plans altogether. Medicare Advantage plans will have to carefully balance the need to reduce medical expenditures and maintain their provider networks to attract enrollment. Establishing strong partnerships with provider systems will be more important than ever.
Q: What do plans need most right now?
Courtney: This is where integrated strategy and actuarial and policy expertise really matter. HMA is supporting stakeholders with payment impact modeling, scenario analysis, and advisory services tied to benchmark rebasing, risk adjustment, Star Ratings, product strategy, and Part D payment policy, so clients can translate the Notice into concrete bid and operating decisions.
From Wakely’s side, the detailed benchmarking and methodology interpretation helps clients quantify what CMS’s technical updates mean in dollar terms and across geographies.
The CY 2027 Advance Notice is also a reminder that average impacts hide portfolio impacts. The plans that model “where the change hits” (diagnosis sources, provider channels, county mix, Stars trajectory) will be best positioned heading into April’s final Rate Announcement.
Blum: And from a policy lens, plans need to connect the dots. CMS’s proposed rate notice is both an articulation of its current priorities and continued progression toward more payment accuracy, encounter-linked data, and program integrity. Medicare Advantage plans should be both prepared to operationalize these policies and to work with the agency to ensure its policies better serve Medicare beneficiaries.
Medicare Advantage plan leaders will be those organizations that operationalize these policy directions early, constructively engage in the policy process, and form far stronger partnerships with health care providers.
You can find more insights on the important proposed changes in plan payments, risk adjustment, and other financial and regulatory requirements for 2027 in Wakely’s summary analysis, Advance Notice of Methodological Changes for CY 2027 MA Capitation Rates and Part C and Part D Payment Policies.

Outlook 2026: Medicare Advantage Advance Notice—What It Means for the 2027 Market

This webinar was held on March 11, 2026 at 12pm ET.
This webinar offered a timely, strategic overview of the recently released proposed 2027 Notice of Benefit and Payment Parameters and what it signals for the evolving coverage landscape. We unpacked the key policy changes, operational requirements, and market assumptions embedded in the rule, with a focus on implications for the individual and small group markets beginning in 2026 and extending beyond.
Participants gained insight into how proposed updates may affect plan design, rate development, risk adjustment, enrollment operations, and health insurance regulation dynamics. We also explored the broader policy direction reflected in the proposal and what organizations should be monitoring now to prepare for implementation and potential downstream impacts.
Learning Objectives:
You can also download the related Wakely Summary of Provisions in HHS’s Proposed 2027 Notice of Benefit and Payment Parameters and Other Key Regulations

On February 11, 2026, the Congressional Budget Office (CBO) released The Budget and Economic Outlook: 2026 to 2036 report. The publication, which represents the first time CBO has released Medicare and Medicaid spending baseline projections since January 2025, reflects the impact of the 2025 Budget Reconciliation Act (P.L. 119-21, OBBBA), recent changes to Medicare reimbursement for skin substitute products, and the latest Medicare Part D and Medicare Advantage bids.
CBO’s baseline serves many functions, including serving as the official “scorekeeping” benchmark used for cost estimates of proposed legislation under consideration in Congress.
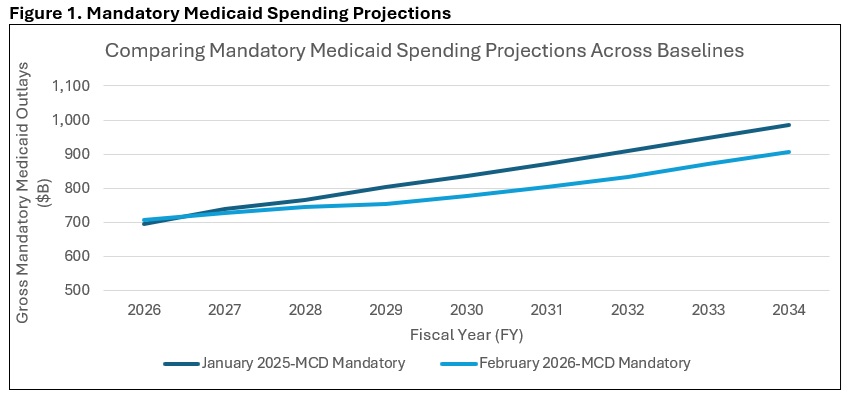
Changes to CBO’s Medicaid Baseline
CBO decreased its projections of 2026–2035 Medicaid mandatory outlays by approximately $514 million from its January 2025 baseline update. The main driver of that reduction is the impact of the Medicaid provisions in the 2025 Budget Reconciliation Act, which CBO expects will reduce total Medicaid enrollment by 13.1 million people in 2035. The drop in Medicaid spending from the OBBBA-related enrollment reductions was partially offset by technical changes CBO made to the Medicaid baseline.
Medicaid costs per enrollee grew by 16 percent in 2025, which was more than CBO had anticipated. The agency attributes the cost per enrollee growth to a reported decrease in the average health status of Medicaid enrollees following the end of the COVID-era continuous eligibility policy.
CBO anticipates that payment rates for Medicaid managed care plans will begin to rise in 2026 because of this decrease in the average health status of enrollees, and the agency has updated the Medicaid baseline accordingly (see Figure 1).
Changes to CBO’s Medicare Baseline
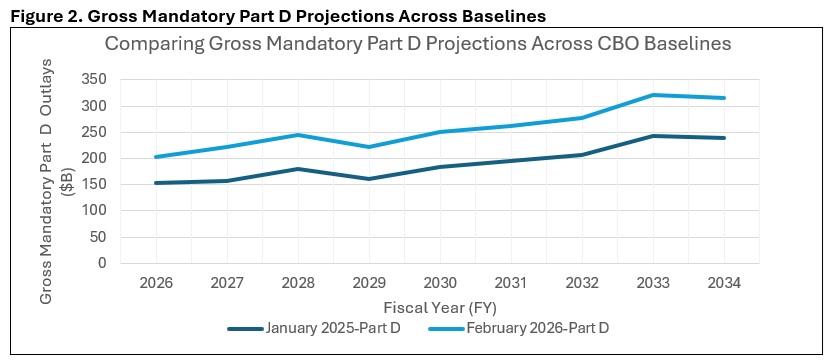
Compared with its January 2025 baseline, CBO increased its projections of Medicare’s 2026–2035 mandatory outlays by about $1 trillion (roughly $942 billion, by Health Management Associates (HMA) calculations). The main driver of that increase came from CBO’s updates to its Medicare Part D spending projections, which were increased to reflect higher than expected 2026 bids from private insurance plans that administer the Part D benefit. According to their 2026 bids, Part D plans anticipate a 35 percent increase in their annual per enrollee costs in 2026—a trend that CBO was not expecting and wants to study further. Part D spending per beneficiary in 2035 is now projected to exceed $4,000, up from less than $3,000 in the January 2025 baseline (See Figure 2).
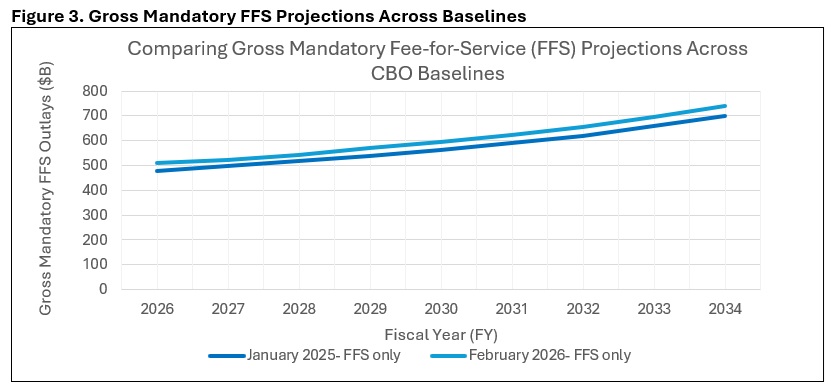
The agency’s Medicare Part A fee-for-service (FFS) spending projection increase was the result of larger than expected increases in 2025 enrollment and per enrollee spending. Those trends were also seen in Medicare Part B FFS but were partially offset by the Centers for Medicare & Medicaid Services’s (CMS) recent reimbursement changes to skin substitute products. Overall, CBO estimates that the skin substitute reform issued in CMS’s Medicare Physician Fee Schedule (MPFS) and Outpatient Prospective Payment System (OPPS) CY 2026 final rules will save $245 billion over the 2026–2035 period, including the effects on the Medicare Advantage (MA) program (see Figure 3).
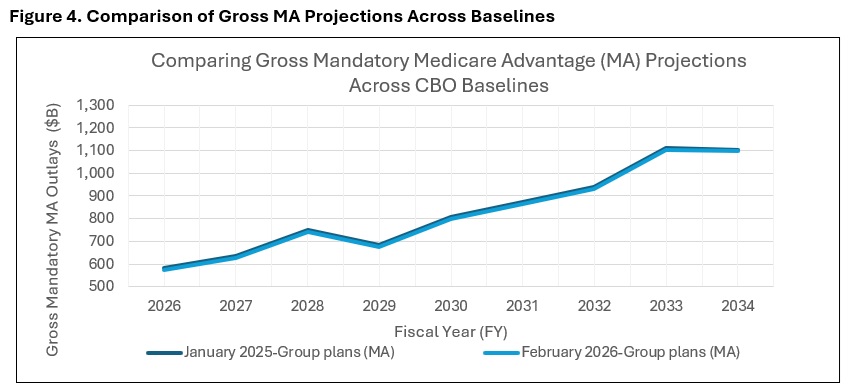
Finally, CBO reduced its spending projections for MA compared to the January 2025 baseline. This change was made to reflect lower-than-expected Medicare Advantage enrollment in 2025, although the spending implications of lower enrollment were partially offset by higher-than-expected bids in 2026 by providers of MA plans (see Figure 4).
Contact an HMA Expert Today
Interested in understanding how CBO’s latest baseline update affects the federal budgetary implications of certain Medicare or Medicaid policy topics or proposals? Contact our experts, Mark Desmaris and Rachel Matthews, to learn more about HMA’s “CBO-style” federal budgetary scoring work, which relies on The Moran Company’s long-standing methodology. [1]
Beyond federal budget scoring, HMA is working with states, health plans, and providers to assess how changes in enrollee health status are affecting utilization, costs, and payment rates—and what those trends may mean for Medicaid and MA organizations and providers. Our teams support states in evaluating managed care rate setting and program design, help Medicaid and MA plans anticipate risk and bid implications, and assist providers in understanding how changes in patient acuity could affect care delivery, contracting, and financial performance.
[1]Specifically, we apply our understanding of CBO precedents to predict how CBO will likely evaluate the budgetary impact of the legislation in question. We use our best judgment to adopt the assumptions CBO would tend to use, with the understanding that any variance in the assumptions CBO ultimately adopts could cause our estimate to differ from theirs.

Updates on Federal Funding for Rural Communities through the Rural Health Transformation Program

The Long View
On December 29, 2025, The Centers for Medicare & Medicaid Services (CMS) announced the highly anticipated funding awards to states for the Rural Health Transformation Program (RHTP)—a five-year, $50 billion federal initiative designed to stabilize and transform rural health systems across the country. This new federal investment marks a pivotal moment for states and their partners to address long-standing challenges in rural healthcare while laying the foundation for broader transformation. It provides an opportunity to reimagine care delivery, strengthen infrastructure, and build sustainable models that address entrenched gaps in rural health.
Directing Resources to Rural Communities
RHTP is designed with a focus on rural communities, where residents face persistent challenges such as provider shortages, hospital closures, and limited access to care. RHTP investments will support infrastructure development, IT system implementations and trainings, workforce recruitment and retention, and innovative care models tailored to rural community needs. The long-term goal is to create lasting capacity and resilience in rural health systems and promote better health outcomes for residents.
But the vision doesn’t stop there.
Catalyzing Statewide Transformation
While rural communities are the primary beneficiaries, we believe the impact of the RHTP will extend beyond rural borders. The program’s design encourages states to develop initiatives that can serve as pilots and start-ups, creating scalable solutions that can be adopted statewide. Workforce development programs, for example, may begin by focusing on rural providers and community health workers (CHWs) and training these individuals but, over time, strengthen the healthcare workforce across entire states and regions.
Much of the federal funding will enable states and their partners to invest in technology modernization, telehealth expansion, and integrated care models. These improvements assuredly will enhance access and quality for rural residents. And these same technologies can be deployed to enhance efficiency and coordination across entire health systems, laying the groundwork for broader system transformation and health improvement. The focus on chronic care management and innovative care arrangements has the potential to improve outcomes for all populations.
Collaborative Pathways for States and Partners
States and their partners—including health systems, community-based organizations, and technology innovators—have a valuable opportunity to collaborate on initiatives. In our review of state applications and the initial wave of state driven funding solicitations, we identified efforts to tackle long-standing system challenges, including:
Looking Ahead
RHTP is more than a funding stream. It is a catalyst for innovation and collaboration, providing an important avenue to address the chronic inequities in quality, access, and outcomes that people living in our nation’s rural communities often experience. But it also could foster improvement statewide. Program evaluation and performance monitoring of the small, community-based programs and the large-scale, multi-site, multi-year initiatives will provide insights that inform strategic decision-making at the local, state and federal levels. By scaling effective rural health-focused initiatives and investing in new and feasible tools, strategies, and programs, states can create models that improve care delivery for all their residents in the future. This is a moment for states, providers, and partners to think big and design programs that deliver lasting impact.
Health Management Associates (HMA) offers support to state agencies, health systems, and community partners shaping rural-first pilots that are designed for scalability—from maternal and perinatal care networks, EMS community care models, caregiver and CHW pipelines, to telehealth modernization and behavioral health integration. Our rural expertise and our unique ability to combine expertise in clinical, operational, policy, and data reforms for care improvement are well-suited to the goals of RHTP.
With the RHTP funding advancing to state partners early in 2026 and annual recalculations of state awards tied to performance, the time to design rural pilots that become statewide programs is now.
For questions about the RHTP opportunities for your organization and the solutions HMA can tailor to meet the needs of your state, contact Kathleen Nolan and Andrea Maresca.
Connecting the Dots: A new blog series for 2026
Connecting the Dots is a monthly HMA blog series that brings together insights from our experts to examine the major policy, program, and market forces shaping healthcare coverage, delivery systems, and financing in 2026. The posts look beyond individual changes, instead connect emerging developments across programs and markets to help leaders understand what’s changing, why it matters, and how their decisions shape the path ahead.

The Centers for Medicare & Medicaid Services (CMS) issued a memo January 27, 2026, with updates on the agency’s approach to checking whether Medicare Advantage (MA) plans are being paid correctly. These reviews are conducted through Risk Adjustment Data Validation (RADV) audits, which help CMS confirm that the diagnoses MA plans report are supported by medical records.
The January 2026 memo signals that CMS intends to honor its commitment to strengthen oversight of MA payments, including accelerating and expanding the use of RADV audits and using AI (artificial intelligence) to streamline human coding reviews. MA organizations must now prepare to respond to the RADV audit notice within the required five-month window, while balancing their other risk-adjustment programs.
In this article, we explain the rapidly evolving landscape affecting RADV audits. Wakely, an HMA company, addresses what these changes mean for MA organizations and key considerations to ensure they are prepared for the upcoming enhancements to federal program integrity initiatives.
Overview of CMS’ RADV Refresh
CMS announced a major shift in May 2025: All MA plans will undergo RADV audits—not just a small sample as before. These audits look for cases in which diagnosis information submitted by a plan does not match the documentation in the patient’s medical record. When this happens, CMS may decide the plan was overpaid and require repayment. Historically, CMS audits have identified widespread diagnosis-code documentation errors, resulting in significant revenue recoupment from MA plans.
The 2025 announcement creates a framework for additional risk for MA plans, which could shift to risk-bearing provider groups. As we explained in an earlier article, key components of that announcement include:
Both the use of extrapolation and the removal of the FFS adjuster were later challenged in court.
Legal Challenge
In September 2023, Humana sued CMS in federal court, arguing that the 2023 RADV final rule, which allowed extrapolation and removed the FFS adjuster, was put into place without following proper federal rulemaking procedures. On September 25, 2025, the court agreed with Humana and vacated certain parts of this final rule, meaning certain parts of the rule are no longer in effect.
CMS appealed the ruling on November 1, 2025, which has created uncertainty about how RADV audits will work in future years.
Navigating the Legal and Regulatory Changes in Early 2026
The court did not say that extrapolation or elimination of the FFS adjuster is illegal—only that CMS did not follow the required process for changing the rules. Hence, the 2023 RADV final rule cannot take effect unless CMS wins its appeal or reissues the policy using the proper steps.
In its January 2026 Health Plan Management System (HPMS) memo, CMS stated that it will comply with the order while it is in effect.
The pending litigation does not diminish CMS’s broader commitment to increased audit activity and heightened scrutiny of MA risk-adjustment practices.
Effect of the Ruling. During RADV audits, CMS selects a sample of enrollees and requests corresponding medical records from the MA plan. These records are reviewed to confirm that the documented diagnoses meet CMS requirements. If unsupported diagnoses are found, CMS may recalculate payments and recover overpayments from the health plan. This audit process maintains program integrity and ensures accurate payments.
Plans that submit incomplete records could owe significant repayments to CMS.
CMS’s January 2026 memo clarifies how the agency plans to roll out additional RADV audits starting with PY 2020. CMS also addresses the agency’s plans to:
Preparing for What’s Next
Given CMS’s stated direction and the still unsettled litigation environment, MA plans should remain vigilant and audit ready.
Key steps include:
Connect with Us
Wakely assists plans with their RADV initiatives and development of robust RADV playbooks. For more information about Wakely’s RADV playbooks, contact Debbie Conboy.

This report synthesizes insights from multiple efforts to support the financial sustainability of HealthySteps sites in California, including federally qualified health centers (FQHCs), community clinics (non-FQHCs), private practices, and other settings. Led by the HealthySteps National Office and Health Management Associates (HMA), the technical assistance (TA) elevated challenges, strategies and best practices to achieve sustainability informed by learning collaboratives, individualized TA sessions, and financial modeling exercises. This report complements additional resources that the HS National Office and HMA developed which are available via the HealthySteps (HS) Sustainability website.

On February 3, 2026, Congress finalized federal funding for fiscal year (FY) 2026, with the House passing the Consolidated Appropriations Act (CAA), 2026, with a vote of 217-214, following Senate approval last week. The president signed the CAA (H.R. 7148) shortly thereafter. The law provides full-year appropriations for the Departments of Health and Human Services (HHS), Housing and Urban Development, Labor, and several other departments.
This year’s HHS funding bill is notable not only for what it includes, but also for what it omits. It restores or maintains funding for key public health and research agencies previously proposed for elimination in the president’s FY 2026 budget request, extends several healthcare programs, and contains a significant package of pharmacy benefit manager (PBM) reforms. All of this activity comes as the Administration announces new grant programs and policy efforts related to its signature priorities.
In this article, we review the major funding and policies approved in the HHS spending bill. We also address key considerations for healthcare organizations as they anticipate downstream funding and policy developments and develop advocacy initiatives for federal FY 2027 bills.
HHS Funding Levels and Direction
The bill provides $116.8 billion for HHS, an increase of $210 million over FY 2025, and rejects large-scale structural reorganizations proposed in the president’s FY 2026 budget. This provision preserves funding for the Agency for Healthcare Research and Quality (AHRQ), Centers for Disease Control and Prevention (CDC), Health Resources & Services Administration (HRSA), and the Substance Abuse and Mental Health Services Administration (SAMHSA)
Table 1. HHS Agency Funding Highlights, FY 2026
| Agency | FY 2026 Funding | (+/-) Compared with FY 2025 |
| Administration for Strategic Preparedness and Response (ASPR) | $3.7 billion | +$58 million |
| CDC | $9.2 billion | level funding |
| Centers for Medicare & Medicaid Services (CMS), administrative expenses only | $3.7 billion | level funding |
| HRSA | $8.9 billion | +$415 million |
| National Institutes of Health (NIH) | $48.7 billion | +$929 million |
| SAMHSA | $7.4 billion | +$65 million |
The bill also extends mandatory funding for community health centers, special diabetes programs, the National Health Service Corps, and Teaching Health Center Graduate Medical Education.
PBM Reforms in the Package
In one closely watched area of federal policymaking, the FY 2026 package includes a substantial set of PBM-related reforms that largely mirror the bipartisan package negotiated but not enacted in December 2024. These reforms have implications across Medicare Part D, commercial insurance, and employer-sponsored plans.
The legislation contains the following PBM reforms:
These provisions position 2026 as a consequential year for PBM regulation, increasing transparency, strengthening plan leverage, and heightening HHS oversight.
Healthcare Extenders and Program Reauthorizations
The bill includes a broad set of Medicaid, Medicare, and public health program extenders, affecting providers, patients, states, and managed care plans.
Medicaid
Medicare
Congress extends several key programs and payment provisions, including:
Behavioral Health Policy
The appropriations bill was finalized as the administration announced new funding and policy initiatives to support behavioral health, crisis services, workforce expansion, and youth mental health—efforts mirrored in SAMHSA’s increased appropriations.
SAMHSA’s $7.4 billion budget includes:
Considerations for Stakeholders
Federal funding and policy developments affect state budget dynamics as many states are now releasing 2026–2027 budget proposals as well as the operational and growth plans of healthcare organizations and partners.
A few key takeaways from the FY 2026 funding bill include:
Connect with Us
If you would like deeper analysis or state and stakeholder-specific effects, HMA’s policy experts are available to assist.