This week's roundup:
- In Focus: Five Takeaways from the CMS Medicaid Managed Care Final Rule
- In Focus: Analysis of Five Key Proposals in CMS’s FY2025 Medicare Hospital IPPS Rule
- Connecticut Lawmakers Plan to Revisit Medicaid Managed Care for Enrollees
- Florida Receives Eight Intents to Protest Against SMMC Awards
- Georgia Governor Signs Bill to Relax Certificate-of-need Regulations, Form Health Commission
- Maryland Total Cost of Care Model Improved Medicare Spending, Decreased Health Disparities in First Four Years
- Massachusetts Receives Federal Approval to Provide Medicaid-funded Temporary Housing for Emergency Shelter Program
- Michigan Medicaid to Offer Maternal Health Quality Payments
- New York Enacted Fiscal 2025 Budget Invests in Health Equity, New Policies Promoting Access to Care
- New Mexico Extends Medicaid Coverage for Seniors Receiving Long-term Care, Individuals with Disabilities
- Texas Lawmakers Urge Governor to Cancel, Delay STAR, CHIP Awards
- HHS Launches Investigation Examining PBMs
- Change Healthcare’s Patient Personal Data Still in Jeopardy
In Focus
Five Takeaways from the CMS Medicaid Managed Care Final Rule
This week, our In Focus section reviews significant Medicaid policy announcements from the Centers for Medicare & Medicaid Services (CMS). For example, both the Medicaid and Children’s Health Insurance Program Managed Care Access, Finance, and Quality Final Rule (CMS-2439-F) (CMS fact sheet available here) and the separate Ensuring Access to Medicaid Services Final Rule (CMS-2442-F) (CMS fact sheet available here) were released April 22, 2024.
Taken together, these two final rules create new flexibilities and requirements aimed at enhancing accountability for improving access and quality in Medicaid and the Children’s Health Insurance Program (CHIP) across the fee-for-service and managed care delivery systems and provide targeted regulatory flexibility in support of this goal.
HMA’s April 11, 2024, “What to Watch For” article outlined several proposed changes that CMS was poised to advance in the Medicaid managed care program. We focus today on the approved changes, including:
- In lieu of services and settings (ILOSs)
- The Medicaid and CHIP quality rating system (MAC QRS)
- Medical loss ratios (MLRs)
- Network adequacy
- State directed payments (SDPs)
Following are HMA’s insights on the key takeaways in each of these major areas for states, managed care organizations (MCOs), providers, and other stakeholders. In addition, HMA experts will discuss the final rule during a LinkedIn Live on event at 2:00 pm (EDT) April 25, 2024. Go to the HMA LinkedIn feed to watch.
In future weeks, HMA will review the Ensuring Access to Care final rule.
ILOSs
The final rule makes clear that CMS remains committed to the conviction that ILOSs can play an important role in supporting state and MCO efforts to address many of the unmet physical, behavioral, developmental, long-term care, and other enrollee needs. At the same time, CMS continues to put forward requirements in this area to ensure adequate assessment of these substitute services and settings in advance of approval, ongoing monitoring for sufficient beneficiary protections, and financial accountability for related expenditures.
The final rule presents an opportunity to leverage ILOSs to improve population health, reduce health inequities, and lower total healthcare costs in Medicaid and CHIP, including by addressing unmet health-related social needs as well as through other avenues. To take full advantage of this opportunity, states and MCOs must ensure that that they are prepared to meet the accountability measures outlined in the final rule and partner with existing providers and community-based organizations that already provide such services and settings.
Medicaid and CHIP Quality Rating System
CMS finalized most proposed provisions related to mandatory quality measures, the process used to update these measures, the ability of states to include additional measures, and the ability of states to apply an alternative QRS if desired. On this last point, CMS is making several modifications to its MAC QRS proposal to clarify the scope of and to reduce the implementation resources needed for an alternative MAC QRS if a state elects to implement one.
States will be required to collect from MCOs the data necessary to calculate ratings for each measure and ensure that all data collected are validated. This will require MCOs to assess their capability to produce the mandated data upon request by states and, to the extent possible, to assess baseline performance on measures and proactively operationalize strategies to improve performance where necessary.
Medical Loss Ratios
The final rule aligns Medicaid and CHIP MLR QIA reporting requirements with the private market to ensure that only those expenses that are directly related to healthcare QIAs are included in the MLR numerator. CMS notes that this provision will allow for better MLR data comparisons between the private market and Medicaid and CHIP markets as well as reduce administrative burden for MCOs participating across these markets.
MCOs will need to model the impact of QIA expenditures that are no longer available for inclusion in the MLR numerator to ensure that a resulting failure to meet any minimum MLR requirements can be avoided, and, if it is projected to occur, a strategy can be developed and executed to avert the problem. CMS made this requirement effective as of the effective date of the final rule with no delay because it believes it is critical to the fiscal integrity of Medicaid and CHIP, adding urgency to MCO compliance action here.
Network Adequacy
The final rule makes clear that CMS has been persuaded that it needs to increase oversight of network adequacy and overall access to care through a new quantitative network adequacy standard. To measure network adequacy, the agency intends to implement wait time standards, complemented by secret shopper surveys to support enforcement.
Wait time standards and secret shopper surveys present opportunities for states, MCOs, and providers to collaborate to enhance access where needed and ensure compliance with the final rule. Undertaking secret shopper surveys ahead of implementation of the wait time standards (effective the first rating period beginning on or after three years after the effective date of the final rule) to determine the current performance relative to maximum wait times is a proactive step that is worth consideration by states and MCOs and can also be employed to foster dialogue with providers to address any areas of concern identified.
State Directed Payments
CMS is adopting its proposal in the final rule to use the average commercial rate as a limit for SDPs for inpatient and outpatient hospital services, nursing facility services, and professional services at academic medical centers. CMS believes that this approach represents a reasonable limit that is supportive of appropriate fiscal guardrails, while still affording states the flexibility to achieve SDP policy goals. States and providers will need to account for this requirement, along with others, as SDPs are developed going forward.
Connect with Us
HMA is ready to support your efforts to understand and take action to account for the managed care final rule’s effects on your state’s or organization’s strategy and operations. Please reach out to [email protected] to connect with our expert team members on this vital set of issues.
Analysis of Five Key Proposals in CMS’s FY2025 Medicare Hospital IPPS Rule
Our second In Focus section reviews the policy changes proposed by the Centers for Medicare & Medicaid Services (CMS) on April 10, 2024, for the Fiscal Year (FY) 2025 Medicare Hospital Inpatient Prospective Payment System (IPPS) and Long-Term Acute Care Hospital (LTCH) Proposed Rule (CMS-1808-P). This year’s IPPS Proposed Rule includes several policy changes that will alter hospital margins and change administrative procedures, beginning as soon as October 1, 2024.
We highlight five proposed policies that are likely to have the greatest impact on Medicare beneficiaries, hospitals and health systems, payors, and manufacturers:
- Annual inpatient market basket update
- New technology add-on payments (NTAP) policy changes
- Transforming Episode Accountability Model (TEAM)
- Hospital wage index and labor market adjustments
- Revision to housing-related diagnosis coding
Stakeholders have until June 10, 2024, to submit comments to CMS on the contents of this regulation and request for information.
Market Basket Update
Proposed rule: Overall CMS’s Medicare 2025 Hospital Inpatient Proposed Rule will increase payments to acute care hospitals by an estimated $3.2 billion in 2024−2025; however, recent trends in economy-wide inflation may alter this estimate by the time the agency releases the final regulation in August 2024.
HMA/Moran analysis: CMS’s 2.6 percent increase is based largely on an estimate of the rate of increase in the cost of a standard basket of hospital goods—the hospital market basket. For beneficiaries, this payment rate increase will lead to a higher standard Medicare inpatient deductible and increase out-of-pocket costs. For hospitals and health systems, payors, and manufacturers the proposed payment increase (2.6%) falls below economywide inflation over the past year (3.5%) and below what Medicare Advantage plans will receive for 2025 (3.7%).1,2 Importantly, based on our expertise with the calculation of the hospital market basket, we anticipate the proposed 2.6 percent increase will increase slightly by the time rates are finalized later this year.
New Technology Add-on Payments (NTAPs)
Proposed Rule: CMS proposes three changes to the NTAP program and discusses NTAP applications for FY 2025:
- CMS proposes to shift the date used to determine whether an otherwise qualifying product is within its newness period. As proposed, if the product’s three-year anniversary occurs after the beginning of the fiscal year on October 1, the product will receive NTAP payments that year.
- CMS proposes to allow products with a hold on their FDA marketing authorization application to be considered eligible for NTAP.
- Beginning with applications approved in the current FY 2025 cycle, the NTAP add-on percentage for gene therapies treating sickle cell disease would increase to 75 percent.
HMA/Moran Analysis: The first two proposed changes are in response to concerns about more restrictive application requirements finalized last year. When CMS shifted the FDA approval deadline to May 1 last year, commenters noted that fewer products would be eligible to receive NTAPs in their third year of the newness period. Allowing all products with a third anniversary that falls within a fiscal year (rather than only those with expirations in the second half of the fiscal year) to receive NTAPs narrowly addresses this concern. More products will qualify for NTAPs during their third year of newness, but that does not necessarily mean that more products will receive three years of NTAPs.
The second proposal tweaks last year’s change requiring a “complete and active” FDA application at the time an NTAP application is submitted to ensure that NTAP applications were far enough along in the FDA review process that information about the product would be available to the public and for CMS staff review. CMS proposal acknowledges that the original bright line rule may have inappropriately excluded potential applicants.
Finally, CMS’s proposal to increase the NTAP percentage for gene therapies treating sickle cell disease aligns with the Cell and Gene Therapy Access Model’s focus on sickle cell therapies. Of note, CMS seeks comment on whether the increased NTAP percentage should be applied only to applicants that have entered value-based purchasing agreements or are “otherwise engaging in behaviors that promote access to these therapies at lower cost.” CMS seems willing to increase NTAP payments in limited situations to boost selected policy goals, but the proposals in this regulation do not represent widespread NTAP payment increases.
Transforming Episode Accountability Model (TEAM)
Proposed Rule: CMS proposes to establish a new mandatory episode-based CMS Innovation Center model, Transforming Episode Accountability Model (TEAM). In the TEAM model, selected acute care hospitals would coordinate care for people with traditional Medicare who undergo one of the five specified surgical procedures:
- Lower extremity joint replacement
- Surgical hip femur fracture treatment
- Spinal fusion
- Coronary artery bypass graft
- Major bowel procedure
Hospitals in the model will assume responsibility for the cost and quality of care from surgery through the first 30 days after the Medicare beneficiary leaves the hospital. Hospitals also must refer patients to primary care services to support optimal long-term health outcomes.
In a first of its kind program, CMS has created a voluntary decarbonization and resilience initiative through which participating hospitals can report metrics related to greenhouse gas emissions to CMS. CMS will provide individualized feedback reports and public recognition of participation and potential performance in the initiative.
HMA/Moran Analysis: The critical aspect of the TEAM model that stakeholders need to understand is that it will be mandatory. TEAM will begin in 2026 and continue for five years. The TEAM model builds on and combines previous models such as the Bundled Payment for Care Improvement (BPCI) model and the Comprehensive Care for Joint Replacement (CJR) model. Hospitals will be required to report various quality measures, and payment will be based on spending targets and include retroactive reconciliation. TEAM also seeks to integrate specialty and primary care. The model complements existing accountable care organization (ACO) models such as ACO REACH or the Medicare Shared Savings Program as beneficiaries would be able to be assigned to both TEAM and ACO programs.
Hospital Wage Index Adjustments and Labor Market Changes:
Proposed Rule: CMS proposes two wage index policies for FY 2025. First, CMS proposes to extend the temporary policy finalized in the FY 2020 IPPS/LTCH PPS final rule for three additional years to address wage index disparities affecting low-wage index hospitals, which includes many rural hospitals. Second, as required by law, CMS proposes to revise the labor market areas used for the wage index based on the most recent core-based statistical area delineations issued by the Office of Management and Budget (OMB) based on 2020 Census data.
HMA/Moran analysis: The two wage index policies that CMS proposes for FY 2025 will have important positive and potentially negative consequences for hospital payment. The policy to extend the low-wage index policy for three additional years will allow many hospitals with low wage indexes to increase their wage index and their payment rates across all MS-DRGs. This policy will bring millions of additional dollars to rural hospitals in FY 2025.
The second policy is a statutorily required update to the labor markets used to establish CMS’s hospital wage indexes. CMS will redefine 53 counties from urban to rural and 54 counties from rural to urban, which will disrupt various hospital payment policies for hospitals in the affected counties. The overall impact of both proposed geographic policy changes for FY 2025 will be to increase inpatient payment rates for rural hospitals.
Revision to Housing-Related Diagnosis Coding
Proposed Rule: CMS proposes to change the severity designation of the seven ICD-10-CM diagnosis codes that describe inadequate housing and housing instability from non-complication or comorbidity (non-CC) to complication or comorbidity (CC).
HMA/Moran Analysis: In proposing this change, CMS is building on its previous policy of including diagnosis codes for describing when a beneficiary is homeless (e.g., unspecified, sheltered, unsheltered). Importantly, this new policy proposal will enable hospitals to be paid higher inpatient payment rates when patients with inadequate or unstable housing are served. Specifically, this proposal would result in cases involving patients to whom these codes apply to be coded in a higher-level MS-DRG within a given family of MS-DRG codes. If finalized, this change in coding policy will result in higher payment rates for hospital patients who are experiencing housing insecurity.
Connect with Us
HMA’s Medicare Practice Group, including consultants from The Moran Company, works to monitor legislative and regulatory developments in the inpatient hospital space and to assess the impact of inpatient payment, quality, and policy changes on the hospital sector. Our Medicare experts interpret and model inpatient policy proposals and use these analyses to assist clients in developing their strategic plans and commenting on proposed regulations. We replicate the methodologies CMS uses in setting hospital payments and model alternative payment policies using the most current Medicare (100%) claims data. We assist clients with modeling for DRG reassignment requests and to support NTAP applications. We also support clients in analyzing CMS Innovation Center alternative payment models.
For more information or questions about the policies described above, contact Zach Gaumer ([email protected]), Amy Bassano ([email protected]), Kevin Kirby ([email protected]) or Clare Mamerow ([email protected]).
HMA Roundup
Alaska
Alaska Releases Draft Behavioral Health Organization Services RFP. The Alaska Department of Health, Division of Behavioral Health released on April 19, 2024, a draft request for proposals (RFP) for a statewide behavioral health services organization to partner in fully implementing services available under the Alaska Substance Use Disorder and Behavioral Health Program Medicaid Section 1115 demonstration waiver. The state submitted an 1115 waiver renewal application to continue this demonstration for another five-year period through December 31, 2028. All stakeholder feedback for the RFP must be provided by May 20. The contract will run from the date of award for three years, with five one-year renewal options. Read More
Colorado
Colorado House Passes Bill to Expand Medicaid Funding of Housing, Nutrition Services. Colorado House Democrats reported on April 22, 2024, that the Colorado House passed a bill, sponsored by Representative Shannon Bird (D-Westminster) and others, that would direct the Colorado Department of Health Care Policy & Financing to conduct a feasibility study and apply for a Section 1115 demonstration to expand Medicaid funding for secure housing and nutrition services. The bill next heads to the Senate for review. Read More
Colorado Submits Section 1115 Substance Use Disorder Demonstration Amendment. The Centers for Medicare & Medicaid Services announced on April 18, 2024, that Colorado submitted a request to amend its Expanding the Substance Use Disorder Continuum of Care Section 1115 demonstration. The proposed amendment will include prerelease services for adults and youth transitioning from correctional facilities, reimbursement for acute inpatient and residential stays for individuals diagnosed with a serious mental illness or serious emotional disturbance, and continuous eligibility for children 0-3 years and 12 months of continuous coverage for individuals leaving incarceration. Public comments will be accepted through May 18. Read More
Connecticut
Connecticut Lawmakers Plan to Revisit Medicaid Managed Care for Enrollees. CT News Junkie reported on April 17, 2024, that some Connecticut lawmakers are interested in revisiting a capitated managed care system for the state’s Medicaid enrollees after the current legislative session adjourns on May 8. The Council on Medical Assistance Program Oversights, which advises the state on its Medicaid program, will discuss the issue at its next meeting. Read More
Florida
Florida Receives Eight Intents to Protest Against SMMC Awards. Health Payer Specialist reported on April 19, 2024, that Aetna, AmeriHealth Caritas, Florida Community Care, ImagineCare, Molina Healthcare, Sentara Care Alliance, South Florida Community Care Network/Community Care Plan, and UnitedHealthcare have notified the state of their intent to protest the contracts awarded for the Florida Statewide Medicaid Managed Care (SMMC) program. Current contractors CVS/Aetna, Molina Healthcare, UnitedHealthcare, and AmeriHealth Caritas did not secure contracts nor did new bidders Sentara Care Alliance and ImagineCare, a joint venture between CareSource and Spark Pediatrics. The state awarded contracts to Florida Community Care, Humana, Elevance/Simply Healthcare Plans, South Florida Community Care Network/Community Care Plan, and Centene/Sunshine State Health Plan. Florida is required to resolve the protest within seven business days or hold a hearing within 30 days if an agreement is not reached. Read More
Georgia
Georgia Governor Signs Bill to Relax Certificate-of-need Regulations, Form Health Commission. The Georgia Recorder reported on April 19, 2024, that Georgia Governor Brian Kemp enacted a bill that would scale back certificate-of-need regulations and form a nine-member comprehensive health coverage commission. The bill will also make it easier to open psychiatric and substance use disorder inpatient facilities and rural hospitals. Read More
Illinois
Illinois Senate Passes Bills to Establish Term Limit for Marketplace Director, Subcommittee to Streamline Medicaid Services. Health News Illinois reported on April 23, 2024, that the Illinois Senate passed a bill, sponsored by Senator Laura Fine (D-Glenview), that would set a two-year term limit for the director of the state’s Marketplace, set to launch in 2026. Governor JB Pritzker has selected Morgan Winters to serve as director and the bill allows the director to serve more than one term. The bill also adds pregnancy as a qualifying life event for a special enrollment period, aligns flat-dollar copay requirements and stand-alone dental plans with federal regulations, and requires insurers exiting the Marketplace to provide 45 days’ notice to the state’s Department of Insurance. The Senate also passed a bill sponsored by Senator Laura Murphy (D-Des Plaines) that would establish a new 30-member Medicaid subcommittee to explore and develop policies to streamline Medicaid services and provider networks for individuals with intellectual or developmental disabilities (IDD). Both bills are currently being reviewed in the House. Read More
Illinois Expects to Receive Federal Approval for SDOH Section 1115 Demonstration Waiver in Summer 2024. Health News Illinois reported on April 22, 2024, that Illinois Department of Healthcare and Family Services (HFS) Director Elizabeth Whitehorn informed legislators that the state expects to receive federal approval of a Section 1115 demonstration waiver, aimed at targeting social determinants of health and behavioral health by this summer. Following approval, HFS will work with federal partners to determine operational protocols and a plan for implementation. Read More
Illinois House Advances Bills for Network Adequacy, Step Therapy Ban. Health News Illinois reported on April 18, 2024, that the Illinois House has advanced a bill which would require insurers to audit at least 25 percent of their provider directories once a year and provide information so consumers may dispute charges for out-of-network providers that were falsely listed as in-network. The state Department of Insurance would also have to audit at least 10 percent of plans annually to ensure network adequacy standards are being met. Separately, the House passed a bill that would ban short-term, limited duration health plans, prior authorization requirements for inpatient mental healthcare, and the use of “step therapy” in prescription drug coverage. Both will move to the Senate for further consideration. Read More
Indiana
Indiana Senior Care Organizations Voice Rate Concerns Ahead of MLTSS Implementation. The Indiana Capital Chronicle reported on April 23, 2024, that Indiana’s senior care organizations are voicing concerns over the state’s transition of Medicaid long-term services and supports to managed care. The sixteen Area Agencies on Aging (AAA) will subcontract with the three managed care organizations (MCOs) selected for the state’s PathWays for Aging program, Elevance/Anthem, Humana, and United. Within the current fee-for-service structure under the Aged and Disabled waiver, AAAs provide case management but will move to provide service coordination for the MCOs, for which the Indiana Family and Social Services Administration previously recommended a 41 percent rate cut. The shift may lead AAAs to lose revenue and choose not to participate in the program, which is scheduled to begin July 1. Read More
Kentucky
Kentucky Governor Signs Bill to Bolster Medicaid Reimbursements for Rural Hospitals. The Appalachian News Express reported on April 18, 2024, that Kentucky Governor Andy Beshear has signed into law a bill to establish a new Medicaid directed payment program for hospitals serving underserved and rural communities. Certain categories of rural hospitals also will be eligible to receive the same Medicaid reimbursements that either the University of Kentucky College of Medicine or the University of Louisville School of Medicine normally receive. If approved by the Centers for Medicare & Medicaid Services, an estimated 43 Kentucky hospitals will benefit from the new plan. Read More
Louisiana
Louisiana Legislature Considers Bill to Improve Maternity Care, Increase Provider Payment Rates. NOLA reported on April 22, 2024, that the Louisiana legislature is considering several bills that would expand health care services to pregnant women and increase payment rates to providers. Specifically, Medicaid patients would receive newborn home visits and remote monitoring between appointments. Another bill would increase payments to physicians who provide obstetric and gynecological care to Medicaid patients. Legislators have also discussed permitting Medicaid coverage for doula care during pregnancy. Read More
Maryland
Maryland Receives Approval for Medicaid Mobile Crisis State Plan Amendment. The Centers for Medicare & Medicaid Services approved on April 15, 2024, a Maryland Medicaid state plan amendment, which will allow the state to develop mobile crisis intervention teams to connect eligible individuals to behavioral health providers 24 hours a day. The program is effective May 1. Read More
Maryland Total Cost of Care Model Improved Medicare Spending, Decreased Health Disparities in First Four Years. The Centers for Medicare & Medicaid Services released on April 8, 2024, a progress report on the Maryland Total Cost of Care model, which examines whether state accountability and provider incentives can improve care and health for Medicare beneficiaries. The report found that the model positively affected spending and utilization in its first four years, from 2019 through 2022. Racial and socioeconomic health disparities decreased, and the Maryland Primary Care Program has improved efficient follow-up for those with chronic conditions. The model aims to save $2 billion in Medicare spending over eight years and meet goals for health care quality and population health. Read More
Maryland Disenrolls 22,220 Medicaid Beneficiaries During March Redeterminations. Maryland Matters reported on April 19, 2024, that Maryland disenrolled 22,220 Medicaid beneficiaries during March eligibility redeterminations, including 17,548 for procedural reasons. More than 300,000 beneficiaries have lost coverage since redeterminations began in March 2023. Read More
Massachusetts
Massachusetts Receives Federal Approval to Provide Medicaid-funded Temporary Housing for Emergency Shelter Program. Mass Live reported on April 22, 2024, that Massachusetts has received federal approval through its Section 1115 demonstration amendment to use Medicaid funds to pay for up to six months of temporary housing for eligible families and pregnant individuals residing in the emergency shelter system. Medicaid will fund $190 million this year and a total of $647.5 million through 2027. There are currently 7,495 families enrolled in the emergency shelter system, which has a 7,500 cap. The request was made in October 2023 when an influx of migrants arrived in the state. Read More
Massachusetts Receives Federal Approval to Expand MassHealth Eligibility, Flexibilities. The Centers for Medicare & Medicaid Services announced on April 19, 2024, that it has approved Massachusetts’ MassHealth Medicaid and Children’s Health Insurance Plan Section 1115 demonstration amendment. The amendment will provide additional health-related social needs services/infrastructure supports; expand Marketplace subsidies to individuals with incomes 300 percent of the federal poverty level (FPL) to 500 percent of the FPL and provide cost-sharing assistance; provide limited services to certain incarcerated individuals for up to 90 days immediately prior to release; and expand continuous eligibility to 12 months for adults over age 19 and to 24 months for beneficiaries age 65 and over who are experiencing homelessness. The extension amendment is effective through December 31, 2027. Read More
Michigan
Michigan Issues Proposed EVV Implementation Policy for Medicaid Personal Care, Home Health Managed Care Programs. The Michigan Department of Health and Human Services issued a proposed policy to require the use of Electronic Visit Verification (EVV) for personal care and home health care managed care providers. Personal care service providers must implement EVV by July 1, 2024. Behavioral health, MI Health Link, MI Choice and Community Transition Services, and Medicaid managed care home health care services programs must implement EVV by September 3, 2024. Programs may use the state EVV system, run by HHAeXchange, or one that still meets state compatibility requirements. Stakeholder comments are due by May 28. Read More
Michigan Medicaid to Offer Maternal Health Quality Payments. The Michigan Department of Health and Human Services announced on April 22, 2024, that it will begin providing payments this fall to hospital birthing units participating in the Michigan Alliance for Innovation on Maternal Health (MI AIM) and in the Joint Commission Maternal Levels of Care (MLoC) Verification Program. The MI AIM quality initiative program seeks to improve maternal safety through the implementation of early recognition patient safety bundles, which comprise of sets of practices meant to improve the quality of care provided during delivery and in the postpartum period. The MLoC program provides hospitals with an independent, external evaluation of their organization’s maternal health expertise, equipment, and resources. The state will also provide hospitals participating in both programs a one-time payment. Read More
Mississippi
Mississippi House Offers Higher Eligibility Limit Compromise for Medicaid Expansion; Senate Remains Stalled. WLBT3 reported on April 23, 2024, that the Mississippi House has offered to raise the income eligibility for Medicaid coverage to 138 percent instead of 100 percent, as part of a compromise on the different House and Senate-approved proposals. Under the House proposal, individuals with income levels up to 99 percent of the federal poverty level would remain under managed care organizations, while individuals between 100 percent and 138 percent of the federal poverty level would remain on the federal insurance exchange. The Senate did not offer any compromise at the public conference committee meetings and no resolution has been made. Read More
New Mexico
New Mexico Extends Medicaid Coverage for Seniors Receiving Long-term Care, Individuals with Disabilities. KRQE reported on April 18, 2024, that New Mexico has received federal approval to provide 12 months of extended Medicaid coverage for seniors receiving long-term care and people with disabilities, aiming to insulate them from disenrollments as the state continues Medicaid unwinding related redeterminations. Eligible individuals must have renewals that were due after April 1, 2023 and were not yet processed, and requires individuals to have an active “Level of Care” on file. The extension applies to programs including the Community Benefit, Developmental Disabilities Waiver, Mi Via Waiver, Supports Waiver, and the Medically Fragile Waiver. Read More
New York
New York Enacted Fiscal 2025 Budget Invests in Health Equity, New Policies Promoting Access to Care. New York Governor Kathy Hochul announced on April 22, 2024, that the fiscal 2025 state budget has been enacted. The budget includes a $7.5 billion investment to promote health equity over the next three years via an amendment to the state’s Medicaid Section 1115 demonstration waiver. Other key highlights include language that would allow the state to implement a Medicaid managed care tax that could result in additional state revenues; legislation to eliminate insulin cost-sharing for state-regulated health insurance plans; legislation protecting New Yorkers earning less than 400 percent of the federal poverty level from medical debt lawsuits; and mandating 20 hours of separate paid sick leave for prenatal care. Read More
Pennsylvania
Pennsylvania House Committee Advances Bill for Medicaid Doula Services. abc27 reported on April 17, 2024, that the Pennsylvania House Health Committee has advanced a bill to allow Medicaid coverage for doula services. The bill would provide Medicaid reimbursement for doula services to pregnant individuals and create a Doula Advisory Board. The bill is pending further consideration in the House. Read More
South Dakota
South Dakota Medicaid Officials Address Expansion Program Enrollment. Public News Service reported on April 18, 2024, that in the first seven months since South Dakota Medicaid expansion launched, 19,811 individuals enrolled out of an estimated 51,000 eligible adults, according to a state report. State officials attribute slow enrollment to expansion rolling out during the start of Medicaid unwinding and anticipate it could take up to two years to reach full enrollment. Read More
Texas
Texas Lawmakers Urge Governor to Cancel, Delay STAR, CHIP Awards. Health Payer Specialist reported on April 19, 2024, that 19 Texas lawmakers are requesting Governor Greg Abbot cancel or delay awards for the State of Texas Access Reform (STAR) and Children’s Health Insurance Program (CHIP) Medicaid managed care contracts, after six locally based health plans were not selected to receive awards. The affected plans are filing protests and considering lawsuits. Read More
Texas Proposed Rule to Require an Adult Be Home While Private Duty Nursing Services Are Provided. The Texas Tribune reported on April 19, 2024, that the Texas Health and Human Services Commission proposed a rule that would require an adult to be present when private duty nursing services are being provided. According to state agency officials the proposed rule clarifies existing policy and does not set new or different expectations about how the policy is monitored or reported. If the rule is implemented, consumer advocacy organizations are concerned it will adversely impact families. Read More
Virginia
Virginia Substance Use Disorders are Underdiagnosed Among Formerly Incarcerated Population. VCU News reported on April 22, 2024, that only 17 percent of Medicaid members who were released from Virginia prisons in 2022 were diagnosed with a substance use disorder (SUD) within three months of release, according to a research study requested by the Virginia Department of Medical Assistance Services. Nationally, it is estimated that 85 percent of individuals who are incarcerated have a SUD. Among Virginia Medicaid members who were formerly incarcerated and diagnosed with an opioid use disorder, about 25 percent received medications for their addiction, compared to 78 percent of all Medicaid members with an opioid use disorder. Read More
West Virginia
West Virginia Completes Medicaid Redeterminations. West Virginia Department of Human Services announced on April 23, 2024, that it has disenrolled 207,674 Medicaid beneficiaries during redeterminations from April 2023 through the end of December 2023. The state renewed coverage for 279,952 beneficiaries. Of those disenrolled, 14,561 individuals have been transferred to the Federal Marketplace to be determined eligible for a plan. The state completed its Medicaid eligibility redeterminations process as of March 2024, although January 2024 through March 2024 data is not yet available. Read More
National
CMS Releases Medicaid Managed Care Final Rule as Part of Trio of Final Rules. The Centers for Medicare & Medicaid Services (CMS) released on April 22, 2024, three final rules that affect financing and access to care in Medicaid managed care and fee-for-service delivery systems. The Medicaid and Children’s Health Insurance Program (CHIP) Managed Care Access, Finance, and Quality Final Rule is intended to improve access and quality to care across managed care plans, strengthens standards for timely access to care and monitoring efforts, enhances integrity standards for state directed payments, and defines the scope of in lieu of services and settings to address health-related social needs. The rule also provides clarity on medical loss ratio requirements and creates a quality rating system for managed care plans. In addition, the Medicaid Ensuring Access to Care final rule establishes minimum payment rate and staffing standards. CMS also released a separate final rule, the Minimum Staffing Standards for Long-Term Care (LTC) Facilities and Medicaid Institutional Payment Transparency Reporting final rule which, establishes minimum nurse staffing standards and require states to report on the percent of Medicaid payments that are spent on compensation for direct care workers and other staff. Read More
CMS to Launch Two Maternal Health Affinity Groups. The Centers for Medicare & Medicaid Services (CMS) announced on April 19, 2024, the formation of two affinity groups under the Maternal and Infant Health Initiative that will be led by Medicaid and Children’s Health Insurance Program staff to address maternal mental health and substance use and improve maternal hypertension and cardiovascular health. A webinar series will also launch and run through the summer of 2024, focusing on pregnant and postpartum challenges while addressing mental health, substance use, and hypertension management. Read More
HHS Launches Investigation Examining PBMs. Health Payer Specialist reported on April 19, 2024, that the Department of Health and Human Services (HHS) Office of Inspector General has launched an investigation into the relationship between health payers’ consolidation and control of pharmacy benefit managers (PBMs) and pharmacies and the price of drugs. The investigation follows reports that some plans pay multiple times more for certain medications than what manufacturers charged for generic versions of the drugs. The U.S. Federal Trade Commission is also currently investigating PBM practices. Read More
Federal Agencies Launch Online Healthcare Antitrust Reporting Portal. Modern Healthcare reported on April 18, 2024, that the Federal Trade Commission (FTC), U.S. Department of Justice (DOJ), and Department of Health and Human Services have created an online portal, HealthyCompetition.gov, to allow the general public to report potentially anticompetitive practices within healthcare. The portal is intended to support efforts to strengthen competition and ultimately lower costs of care and prescription drugs. Complaints will be reviewed by the FTC and DOJ’s Antitrust Division and may result in further action. Read More
Biden Administration Revises 340B Dispute Resolution Process Rule. CQ reported on April 18, 2024, that the Biden Administration finalized a rule that includes a revised dispute resolution process for providers and drug companies participating in the 340B program, which requires drug companies to provide discounts to safety-net providers. The rule is intended to streamline the process and removes the previous requirement for complaints to have a minimum threshold of relief. Read More
Nursing Homes Claim New Staffing Rules are Unreasonable Due to Workforce Shortage. NPR reported on April 24, 2024, that new federal regulations establishing a floor for nurse staffing levels in nursing homes will require these facilities to hire thousands more nurses and aides over several years. The Centers for Medicare & Medicaid Services estimated that the rule will increase costs for nursing home operators by approximately $6 billion. Read More
FTC Chair Claims New Technologies Enable Companies to Engage in Health Care Price Fixing. KFF Health News reported on April 23, 2024, comments from the Federal Trade Commission (FTC) chair Lina Khan that algorithms and new technologies are allowing companies to engage in price fixing without having to coordinate with one another, despite there being no algorithmic exemption to the antitrust laws prohibiting price fixing. The FTC is scrutinizing the use of artificial intelligence and algorithms to set prices for individual consumers based on behavioral data collected. Read More
Industry News
Change Healthcare’s Patient Personal Data Still in Jeopardy. Reuters reported on April 22, 2024, that UnitedHealth Group conducted an initial review of the stolen data from the cyberattack on Change Healthcare and found that the files of a “substantial proportion” of Americans were stolen with protected health information or personally identifiable information, despite the company paying a ransom demand. Another hacker group posted screenshots on the dark web which contained UnitedHealth customers’ protected healthcare and personal data. A full analysis of the breached data is expected to take several months. Read More
Community Health Systems to Sell Tennessee-based Hospital to Hamilton Health Care System. Community Health Systems announced on April 19, 2024, that it will sell its 351-bed Tennessee-based Tennova Healthcare and certain related businesses to affiliates of Hamilton Health Care System for $160 million in cash. The transaction, pending regulatory approvals, is expected to close in the third quarter of this year. Read More
Bridges Health Partners Files Lawsuit Against Aetna Over Medicare Supplemental Benefits. Modern Healthcare reported on April 18, 2024, that Bridges Health Partners, a clinically integrated network in western Pennsylvania, filed a lawsuit against Aetna alleging that it broke contract terms by categorizing Medicare supplemental benefits such as gym memberships and gift cards as medical expenses. The suit, filed in state court, is requesting that Aetna stop passing on the cost of supplemental benefits to its health system partners for the remaining two years of its contract. Read More
Private Equity Backed Healthcare Companies Account for a Fifth of Bankruptcies in 2023. Modern Healthcare reported on April 17, 2024, that private equity-owned businesses accounted for at least 17 out of the 80 bankruptcies of healthcare companies in 2023, with the two largest being KKR Group-owned staffing company Envision Healthcare and cancer-treatment provider GenesisCare. Venture-capital backed companies made up another 12 of the filings, according to a Private Equity Stakeholder Project report that looked at companies with liabilities of more than $10 million. The trend is projected to continue in 2024 as companies are increasingly facing credit rating downgrades and potential defaults. Read More
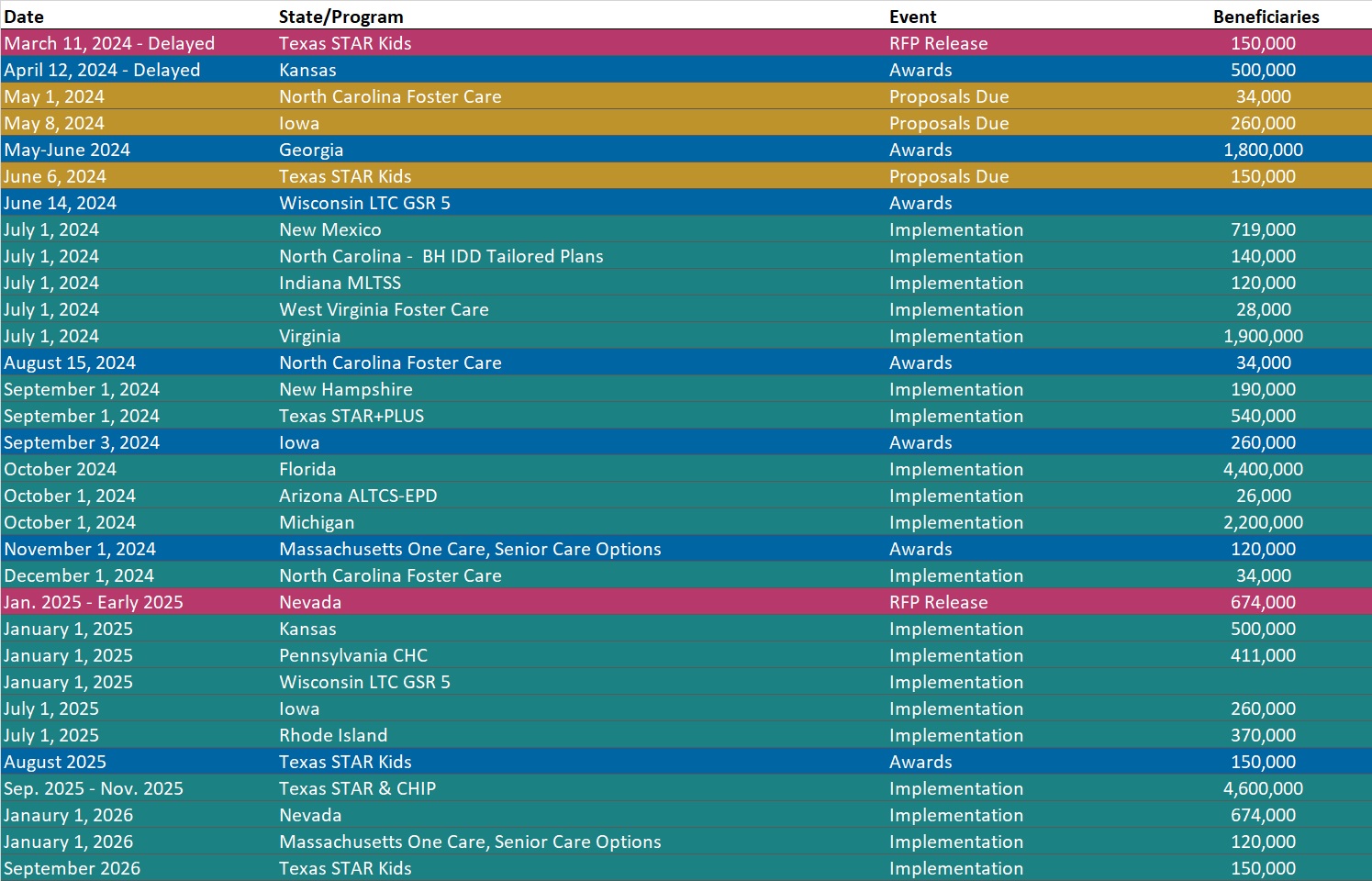
RFP Calendar
HMA News & Events
HMA Webinars:
Substance Use Disorder (SUD) Ecosystem of Care: Building Systems-Thinking in the SUD Ecosystem. Wednesday, May 1, 2024, 12 PM ET. The final webinar of this three-part series emphasizes the importance of a comprehensive and interconnected spectrum of engagement and treatment strategies. To truly build and maintain an SUD ecosystem with accountability across the system and ‘no wrong door,’ best practices must embrace a systems-thinking approach. An interconnected system requires building strong partnerships across the SUD ecosystem and engagement and treatment strategies will focus on leveraging those partnerships to facilitate engagement of individuals throughout the system. Register Here
Managed Care Final Rule LinkedIn Live. Thursday, April 25, 2024, 2PM ET. Policy changes fall into the following major categories: in lieu of services and settings, the Medicaid and CHIP Quality Rating System, medical loss ratios, network adequacy, and state directed payments. These revised policies will affect Medicaid coverage and reimbursement for years to come. HMA experts will highlight their initial takeaways and insights for organizations affected by the new rules. Read More
HMA Podcasts:
Can Data Shape the Future of Medicare’s Value Proposition? Tim Murray is a principal and senior consulting actuary of Wakely Consulting Group, an HMA Company. With over two decades of experience as a health actuary, Tim illuminates the challenges and opportunities within Medicare, particularly focusing on value assessment and the pivotal role of data collection. Digging into the complexities of Medicare Advantage, he discusses predictive modeling, innovative supplemental benefits, and the need for structured data metrics to drive sustainable healthcare solutions. Listen Here