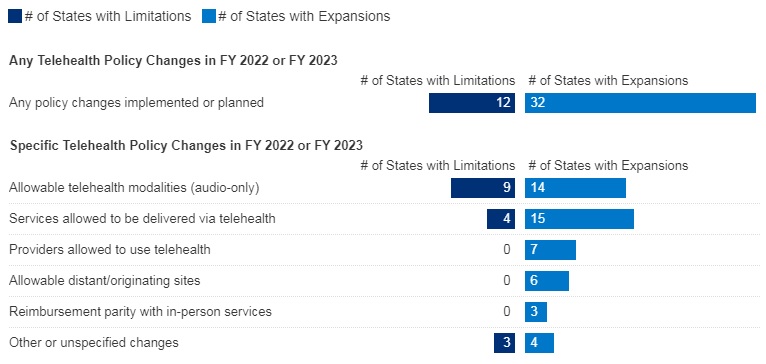
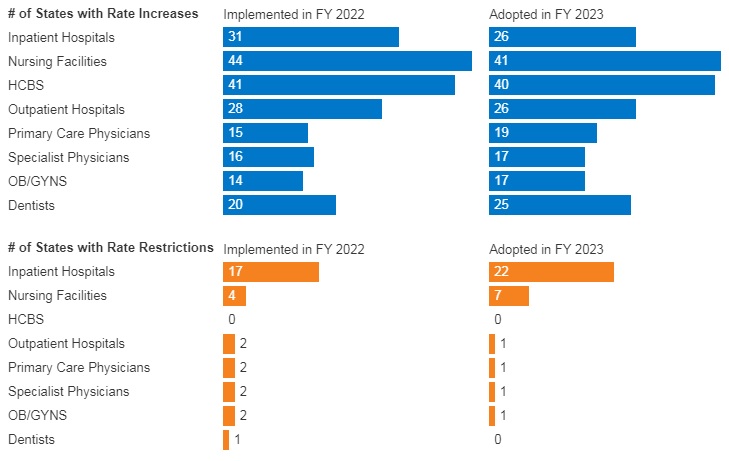
The first quarter of 2026 marked a turning point in federal oversight of pharmacy benefit managers (PBMs), the intermediaries that manage prescription drug benefits for most health plans across the commercial insurance market, Medicare Part D, and other programs. New legislation, agency rulemaking, and enforcement activity collectively signal a new phase of oversight that could materially reshape PBM contracting, compensation, and transparency requirements.
Most notably, the following developments stand out:
- The US Department of Labor (DOL) proposed new disclosure requirements for PBMs that serve self-insured Employee Retirement Income Security Act (ERISA) plans.
- The Consolidated Appropriations Act of 2026 (CAA 26), signed February 3, 2026, establishes comprehensive PBM transparency and contracting requirements in the commercial insurance market and Medicare Part D.
- The Federal Trade Commission (FTC) finalized a settlement with Express Scripts, Inc. (ESI), requiring significant changes to ESI’s business practices.
Together, these actions signal a trend toward greater PBM accountability, with implications for plans, pharmacies, manufacturers, and consumers. This article provides a high-level overview of the major recent developments in the PBM reform policy landscape, along with key considerations for stakeholders.
Medicare Part D: Key Statutory Changes
Beginning in plan year 2028, CAA 26 makes significant changes for PBMs operating in Medicare Part D. Key provisions include:
- Requiring PBMs to provide annual reports to plan sponsor clients detailing aggregate and drug-specific costs
- Restricting PBMs compensation structures, prohibiting payments tied to drug prices, rebates, or price-based benchmarks and limiting PBMs to only receive bona fide service fees that reflect fair market value
- Stipulating additional parameters related to rebate guarantees, contract terminology, and audit rights
Additional provisions that will take effect in beginning with plan year 2029 include:
- A requirement that plan sponsors and PBMs comply with forthcoming standards for “reasonable and relevant” pharmacy contracting terms and conditions
- Expansion of the enforcement infrastructure to avert potential violations of the program’s pharmacy contracting requirements
Key considerations: The Centers for Medicare & Medicaid Services (CMS) has broad discretion in implementing these provisions, including setting pharmacy contracting standards, determining which PBM affiliates are subject to new requirements, and defining “fair market value.” PBMs will face expanded reporting and compliance obligations, while plans and other stakeholders will have opportunities to shape implementation through the regulatory process.
Commercial Health Insurance Market: Key Statutory Changes
For the commercial market, CAA 26 establishes similar transparency requirements for PBMs that serve fully insured and self-insured plans, with reporting required up to four times per year. Unlike Medicare Part D, the statute does not prohibit pricelinked compensation in the commercial market, but it does require detailed disclosure of PBM fees and revenue streams. For contracts with selfinsured plans, PBMs must remit 100 percent of rebates and fees tied to drug utilization, subject to specified limitations.
Key considerations: These provisions significantly expand federal oversight in the commercial market. PBMs will need to scale compliance infrastructure, while employers and other plan sponsors may seek enhanced analytical and actuarial support to interpret disclosures and assess PBM performance.
Medicaid Left Out, For Now.
Unlike prior legislative packages, CAA 26 does not include Medicaid-specific PBM reforms, such as prohibitions on spread pricing (i.e., a PBM charges a payer more than the amount it pays the dispensing pharmacy for a prescription) and expanded National Average Drug Acquisition Cost (NADAC) reporting.
Key considerations: These policies continue to have bipartisan support and could reemerge in future legislation. States, PBMs, and managed care plans should continue monitoring for renewed federal action on these policies.
DOL’s Proposed PBM Fee Disclosure Rule
DOL’s proposed rule, “Improving Transparency Into Pharmacy Benefit Manager Fee Disclosure,” would require PBMs serving self-insured ERISA plans to disclose information about rebates, manufacturer fees, pharmacy payments, and spread pricing. In late February, DOL extended the public comment period to April 15 to allow stakeholders to address how the proposed rule should align with the newly enacted CAA 26 provisions.
Key considerations: DOL could withdraw the proposal in favor of the statutory framework or could finalize the rule to take effect before the CAA 26 requirements begin. Either path would further increase near-term compliance for PBMs and plan sponsors, and stakeholders should monitor this space closely.
FTC Settlement with ESI
The FTC’s settlement with ESI resolves insulin-focused litigation against the PBM and imposes extensive requirements related to transparency, compensation, rebates and fees, and benefit design. The settlement also includes less common provisions, such as a commitment to reshore and increase disclosures related to ESI’s rebate group purchasing organization (GPO) functions.
Key considerations: If similar settlements are reached with other PBMs, the FTC could play an expanded role in shaping PBM market behavior, supplementing legislative and regulatory reforms with enforcement-driven standards.
State Efforts to Regulate PBMs
States continue to pursue PBM reforms, with dozens of laws enacted in recent years addressing licensure, reporting, pharmacy reimbursement, and contracting standards. Although the Supreme Court’s 2020 decision in Rutledge v. PCMA opened the door to certain state-level reforms, subsequent court decisions have narrowed the scope of permissible state regulation, particularly when ERISA preemption or Medicare Part D conflicts arise.
Key considerations: Stakeholders operating across multiple markets and states will continue to face a complex and evolving patchwork of requirements, underscoring the importance of ongoing policy tracking and compliance coordination.
Connect with Us
Recent federal and state actions suggest that PBM reform is entering a more operational phase defined by transparency and enforceable standards governing compensation, contracting, and market behavior. As implementation unfolds, stakeholders across the prescription drug supply chain will need to engage closely with regulators, assess new data flows, and adapt their business practices to a more prescriptive oversight environment.
For more information about the policies described in this article and the PBM policy landscape more broadly, please contact our experts Conor Sheehey or Stephen Palmer.