1260 Results found.

Webinar Replay: How Community Care Hubs Can Enhance Family Caregiver Support Services
This webinar was held on December 3, 2024.
To better support family caregivers and the older adults who they care for, Area Agencies on Aging and other aging network agencies are creating Community Care Hubs (CCH) to address social determinants of health, integrate health and social care, and reduce care costs. In this webinar with LTSS policy experts and providers, we described the implementation of the CCH model in projects in Massachusetts and New York.
Learning Objectives:
- Describe the Community Care Hub (CCH) model for integrating health and social care
- Identify key CCH features to enhance family caregiver support services
- Illustrate the implementation of the CCH model for supporting family caregivers with projects in Massachusetts and New York
Featured Speakers:
- Kristie Kulinski, MSW, Director of Office for Network Advancement, Administration for Community Living
- Nikki Kmicinski, MS, RD, CDH, Chief Executive Officer, Western New York Integrated Care Collaborative
- Jennifer Raymond, JD, MBA, Chief Strategy Officer, AgeSpan

How satisfaction impacts Medicare Advantage plans Star ratings
Medicare Advantage (MA) Star ratings are more than a quality score—they shape the financial and operational success of MA plans. These ratings hinge on factors that every plan can impact by developing continuous improvement processes. The Consumer Assessment of Healthcare and Provider Systems (CAHPS) survey, Healthcare Effectiveness Data and Information Sets (HEDIS) ratings, and the Member Retention rate are all significant levers affecting Star ratings.
The importance of member retention rate
Member retention rate is a measure of member satisfaction but also impacts plan scale. One Medicare Advantage (MA) plan typically reports 0% voluntary disenrollment each year. Another plan is reporting 60% voluntary disenrollment. The voluntary disenrollment threshold is currently set at 18% for a 4-star rating and 10% for a 5-star rating on the measure. The average MA plan is losing more than $60 million in Medicare premium annually due to voluntary disenrollments. The voluntary disenrollment measure excludes members moving out of the service area or sponsor-initiated contractions of the service area.
CAHPS survey impact on Star ratings
CAHPS metrics are an important factor in the Centers for Medicaid and Medicare Services (CMS) Star rating system. MA plans need to develop strong companywide focused member experience processes to help them navigate the healthcare delivery system and community resources available. Evaluating the entire member experience from enrollment through access to care, messaging, outreach, customer service, to disenrollment, involves mapping out every member touchpoint, from a population health approach, to ensure the plan has a caring, approachable, supportive, and balanced experience with the member. Opportunities to eliminate frustrative process steps include identifying health related social needs and disparities that provide easier and time-sensitive access to care and services that are essential to increasing member satisfaction and engagement.
Health plans need a process to identify members who are most likely to be dissatisfied due to events and contact these members to understand the needs and resolve issues quickly. A dissatisfying process issue will repeat if not addressed. Understanding what data the health plan should be continually monitoring and the steps to effectively address any issues is essential to increasing trust with members. It is imperative that members get the opportunity to express their concerns to the health plan with the opportunity to resolve issues satisfactorily before they receive a CAHPS survey.
HEDIS and Star ratings
MA plans need to develop focused processes to proactively monitor HEDIS metrics and drive improvement interventions to keep up with the competition. Having a holistic approach to monitoring, understanding the status and what gaps persist, and a year-round strategy for addressing these gaps is essential to being able to focus efforts on improvement.
As the National Committee for Quality Assurance (NCQA) is moving from a hybrid sampled process to an administrative whole population calculation system, it’s essential that MA plans are addressing each measure in its entirety throughout the year. Digital measurement and Electronic Clinical Data Sets (ECDS) measures are increasing with CMS having a goal of interoperability and implementation of digital quality measures by 2030. Changes with CMS Star metric weightings has increased the total percentage that HEDIS impacts the overall calculation.
Partnering with Pharmacy Benefit Manager to improve Stars
Medicare Part D measures are among the most highly weighted measures in the CMS Stars performance program. Having a strong Pharmacy Benefit Manager (PBM) p artner is a necessity for success. Measures include medication adherence for high blood pressure, cholesterol, and diabetes. Successful plans ensure that members have sufficient prescription fills and re-fills to cover 80% of the days during the year. Measures are scored based on the percentage of members in the denominator who are compliant by the end of the measurement year. Member satisfaction with the plan’s pharmacy program is a key determinant in plan rating by the member and plan retention, impacting other parts of the CMS Stars program, whether Part D is measured alone or as part of an MA-PD plan.
Accelerating Star Rating Performance
The HMA Stars Accelerator Solution offers a comprehensive, results-oriented approach to Star Rating performance improvement that addresses the multifaceted challenges faced by health plans. It examines your plans leadership structure, operational processes, technology, reporting, member-centric engagement, provider partnerships, and develops a strategy for your organization using a data-driven approach for continuous improvement. Multiple “what-if” scenarios are developed that identify top priorities. Measure thresholds that are too far to reach are replaced by measures that are within reach during the final months of the year.
The Accelerator approach includes “all-hands-on-deck” – care coordination, customer service, network development, marketing, analytics, and others. Accelerator plans introduce provider and member incentives and/or fee schedule adjustments to increase interest. These plans also provide information to providers on those attributed members who have measure gaps to facilitate provider outreach that is coordinated with plan outreach.
HMA Accelerator plans experience a reduction in members choosing to leave, attributed more to prevailing cultural changes over time than to enhanced benefits or member rewards. This program is a cultural transformation designed to strengthen star performance. Click here to learn more about the HMA Stars Accelerator Solution’s capabilities, where you can request a copy of the HMA Stars Accelerator Playbook. Let’s have a conversation about how your plan can improve member retention for increased star rating and increased enrollment scale.
Watch a replay of Mastering Star Performance: Strategies from the HMA Stars Accelerator Program.

In behavioral health, parity is essential, but not enough
Today’s post is by Linda Rosenberg, who has recently joined HMA as a Senior Advisor. In this blog she offers her perspective on parity rules for behavioral health from her many years of experience in the field, most recently as the President and CEO of the National Council for Mental Wellbeing until her retirement in 2019 and as part-time faculty member at the Columbia University Department of Psychiatry.
Attending the 2024 Alignment for Progress conference and experiencing the collective commitment to the 90/90/90 goals, I was once again struck by the groundbreaking nature of the Mental Health Parity and Addiction Equity Act of 2008. The legislation was the critical step in ensuring mental health and substance use is treated on equal footing with physical health. Patrick Kennedy, both as the driver of the Act and in his ongoing advocacy helped us to reshape national conversations and policies.
The new regulations released by the Biden administration add much-needed teeth to the Mental Health Parity and Addiction Equity Act. The regulations take on one of the biggest ongoing challenges: the lack of adequate provider networks. Behavioral health clinicians are far harder to find in-network compared to medical providers, with many leaving networks due to low reimbursement rates. Under the new regulations, insurers must maintain adequate networks, regardless of the challenges, which will likely come with significant costs to entice clinicians back.
Implementation of the regulations won’t be simple. The insurance industry is sorting out what compliance will mean to their operations and bottom line. The federal government is struggling to fund and build a monitoring infrastructure. State governments need to understand their roles and responsibilities. And patients and the people who love them need to learn about their expanded rights and how to exercise those rights. Everyone has a job to do.
The intent of the parity law was about ensuring that mental health and addiction services are treated with the same urgency, seriousness, and respect as any other form of medical treatment. And yet parity has remained a promise unfulfilled for too many. The new regulations are a welcome and necessary step forward, but they cannot address all that needs to be done. Parity is essential, but it’s not enough.
Early on in my tenure and long before I retired from the National Council for Mental Wellbeing, a very special member and mentor Carl Clark MD, CEO of WellPower in Denver shared a secret with me. There are “wicked” problems, and wicked problems don’t have a single solution. A wicked problem is complex and interconnected … and has no stopping rule, rather wicked problems are opportunities for progress.
For too long I’ve listened to too many talks and read too many reports about “fixing” or “creating” a behavioral health system, but the reality is far more complex, far more “wicked”. Fragmentation is endemic to all of healthcare in the USA, we have no single healthcare system and no unified behavioral health system either. We have thousands of systems—public, private, nonprofit, hospital-based, and government-run – each serving different populations and communities with varying levels of resources and approaches and each dependent on a bottom line.
The fight for parity was never just about changing laws—it’s about changing hearts, minds, and systems, reshaping the way we understand and deliver care across all these thousands of systems we’ve created and continue to create.
Well intentioned programs with layered initiatives focused on whole health, social determinants of health, and other efforts are adding complexity to a system that’s already overwhelming for the very people these systems are supposed to serve.
What we need is a financing model that ties all the pieces together – Certified Community Behavioral Health Centers (CCBHCs) are a promising start – a financing model that pays for the continuum of services, inpatient and community, rather than the current fragmented approach that pays for pieces separately. At the same time, we need to leverage technology to alleviate pain points, establish desperately needed standards of care, and provide decision support for both clinicians and patients. With technology, we can measure and benchmark care across systems, creating transparency and accountability at every level.
By aligning financing with the full spectrum of services and using technology to drive transparency and accountability, we can finally begin to address the wicked problems that prevent effective mental health and addiction care. As I help non-profits, health technology companies, and venture firms build growth strategies that result in consumer and economic benefits, I understand that the new regulations give us a foundation to build on—the rest is up to us.

Nevada releases Medicaid managed care RFP: State will expand managed care statewide into rural areas
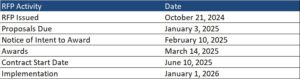
This week, our In Focus section highlights the State of Nevada’s October 21, 2024 request for proposals (RFP), which will expand Medicaid managed care to cover nearly all populations in all counties. The Department of Health and Human Services’ Division of Health Care Financing and Policy (DHCFP) estimates that the expansion statewide will cover 75,000 additional individuals who live in rural areas, including children, parents, and adults without children. The expansion to rural areas in all counties presents new opportunities and critical issues for managed care plans, ensuring that they meet the needs of rural populations effectively.
Background
In 2024, Nevada covers 788,000 Medicaid members, with risk-based capitated Medicaid managed care making up about 75 percent of the total Medicaid population. Managed care covers traditional Medicaid and expansion, the Children’s Health Insurance Program (CHIP) known as Nevada Check Up, and children who have aged out of foster care. Enrollment in an MCO is mandatory for these populations. Currently, Medicaid managed care is only offered in the urban Washoe and Clark counties, which include cities such as Reno and Las Vegas.
Nevada has four MCOs that were procured in 2021: Centene/SilverSummit HealthPlan, Elevance/Anthem, Molina, and UnitedHealthcare/Health Plan of Nevada. These MCOs serve approximately 588,000 beneficiaries in Urban Washoe and Urban Clark counties as of August 2024.
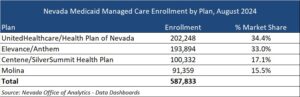
United and Elevance make up the majority of market share by enrollment, with 34.4 percent and 33 percent respectively.
Individuals who receive Medicaid through fee-for-service (FFS) are Medicaid-enrolled children in foster care, juvenile justice, and child welfare systems; individuals with disabilities; seniors; and individuals receiving services through one of the three 1915 home and community-based waiver programs. These individuals will continue to receive services through FFS.
RFP Highlights
The RFP describes the state’s three managed care service areas (SA): Urban Washoe, Urban Clark, and Rural. The Rural SA will include all other counties in Nevada in addition to the rural areas of Washoe and Clark counties.
MCOs must bid on all service areas. DHCFP anticipates selecting four plans. The two awarded vendors with the highest rural care score will operate in all three SAs. The remaining awarded vendors will operate in Urban Clark and Urban Washoe SAs. The state has the option to award a fifth contract to an MCO to operate in the Urban Clark SA only.
The RFP focuses on rural care and policies designed to improve outcomes and access to care, reduce burdens for providers to participate, and simplify administrative tasks for the state. MCOs will need to show their understanding of the unique challenges facing rural providers. They will describe their approach for provider outreach, contracting, and provider training strategies in rural areas, with a focus on primary care, maternal and child health, and behavioral health. Due to the geographic limitations, telehealth will also play a strong role. MCOs will need to address limitations such as access to internet and provide an approach to help members access telehealth. Additionally, MCOs will need to provide their experience in managing non-emergency medical transportation (NEMT) in rural areas and describe an approach for establishing and maintaining a network of transportation providers in these remote areas.
MCOs also will be required to offer at least one Silver and one Gold Qualified Health Plan (QHP) on Nevada Health Link Marketplace by the 2026 coverage year. The state expects this contractual requirement will help reduce churning and improve continuity of care for individuals and families who have a change in eligibility status.
MCOs must also contract with providers that use alternative payment methodologies (APMs), and plans will need to outline value-based purchasing (VBP) strategies within their proposals. APM contracting strategies must support priority areas such as addressing health-related social needs (HRSNs) and improving health equity, access, behavioral health, and maternal and child health outcomes. APM contracting strategies must include quality measures in the payment methodology and outline reporting and estimated financial details. Additionally, MCOs are required to develop a Population Health Program, so proposals must outline how it will leverage specific APMs to meet the program’s goals.
Evaluation
MCOs will require a minimum score of 945 points (out of 1,350 points) on the Technical Proposal to be eligible to win a contract. The Building Provider Networks and Access to Care technical questions is worth the most points, 450, while 300 points are available under the Rural Care and Service Area Expansion section. The table below provides a breakdown of the Technical Proposal Scoring.

The state assigns the highest number of points to the section addressing provider networks and access to care followed by the section addressing rural care and service area expansion.
Timeline
Key Considerations
HMA experts identified the key considerations for MCOs, partners to MCOs, providers who will furnish services to members, and other interested stakeholders.
- The Building Provider Networks and Access to Care Rural Care and Service Area Expansion sections together are worth 750 points, most of the minimum needed, and more than half of the total available points. This is a strong indicator of that MCOs need to demonstrate capacity and innovation to ensure access to members using multiple strategies. Ensuring a robust network of healthcare providers in rural areas is crucial. This includes recruiting and retaining providers who are willing to serve in these regions.
- In this RFP, DCHFP is evolving its value-based payment (VBP) initiatives and expectations for MCOs. The VBP strategies are intended to enhance care quality, improve patient outcomes, and ensure the sustainability of the Medicaid program. MCOs will need to design these models and successfully work with providers to implement the strategies and models.
- Both improving maternal and child health outcomes and addressing mental health and substance use disorders are priorities for the state. These can be more challenging in rural settings due to limited access to specialized care and will require rural specific solutions.
- Changing the Medicaid delivery system model in some rural areas may create the need for building awareness and understanding for the changes that come with this transition. MCOs should be prepared to effectively address the issues of equity and disparities that are present in in rural communities.
Connect with Us
HMA’s experts understand the Medicaid managed care environment and specific issues presented by rural areas. We work with clients to address the multilevel challenges for delivery of quality healthcare and social services to rural populations, and the workforce concerns they create. We identify and help plan for access issues such as lack of services, transportation difficulties, and socio-economic barriers. HMA knows the difficulties that often keep rural providers and organizations from achieving their full potential to serve and support their communities’ need and help Medicaid clients in rural areas in states around the country to solve these difficult problems.
Read more about the work we are engaged in with a range of healthcare industry leaders focused on rural and frontier areas.

October 30, 2024
Nevada Releases Medicaid Managed Care RFP: State Will Expand Managed Care Statewide Into Rural Areas

Healthcare solutions unlocked: Key takeaways from the 2024 HMA Conference
The HMA Conference – Unlocking Solutions in Medicaid, Medicare, and Marketplace – was held in early October in Chicago, drawing a crowd of 350 participants hailing from all parts of the healthcare ecosystem. In the words of one attendee, what made the HMA conference unique was that “it was very rare to have providers, CBOs, health systems, insurers, and public sector organizations in one place…I gained significant value by viewing similar issues from different perspectives.”
Attendees participated in plenary sessions and breakouts that were grounded in Medicaid, Medicare, and Marketplace, with each session extending beyond the traditional topics for these public health insurance programs. Attendees were challenged and inspired to consider the cross-cutting work underway to address health equity, the integration of housing into healthcare, innovative strategies for behavioral health coverage for adults as well as children and families involved in the child welfare system, and the opportunities for federally qualified health clinics to engage in value-based care delivery to improve outcomes.
Signature HMA discussions with health plan leaders and Medicaid directors also provided valuable insights that will help guide the next phase of innovative programs and technologies designed and deployed to improve health. Breakout sessions offered in-depth exploration across Medicaid/Duals, Medicare/MA, and Marketplace/ACA tracks, alongside discussions on demand for innovation, advances in treating sickle cell disease, and creative workforce strategies. The conference concluded with a panel that prepared attendees for policymaking in the post-election, post-Chevron deference landscape.
The HMA event created opportunities to learn and network with potential partners against the background of the city of Chicago skyline, as one participant put it, “away from the daily craziness.” The discussions were robust and focused on new ideas that can be deployed by stakeholders all trying to improve the accessibility, quality, equity, and value of healthcare.
Listed below are conference takeaways that will be of interest and relevant to the broader healthcare ecosystem:
Systemize the little things that improve outcomes.
Keynote speaker Dr. Darshak Sanghavi from ARPA-H challenged people to focus on systematizing the “little things” that drive measurable improvements in outcomes as a source of meaningful innovation. His presentation focused on how ARPA-H is investing differently in private sector innovation – not just big breakthroughs, but also in data-informed approaches that produce consistent quality.
Housing is a healthcare issue.
There is increasing overlap between initiatives to address housing support in Medicare and Medicaid. Bridging diverse sectors and stakeholders is essential to address critical gaps in service delivery. HMA’s new Housing Services & Supports practice group recognizes that housing is part of healthcare, but it does not have to be fully funded by healthcare entities.
Cross-payer collaboration would improve behavioral health.
Behavioral health coverage has historically relied on the Medicaid-based chassis of coverage, but the breadth of needs and federal parity requirements have created an urgent need for new approaches to coverage across all systems and all payers. Cross-payer innovation and collaboration are essential, and systems need to position themselves to scale effective solutions that allow individuals to access services when they need them. In particular, youth and family voices must be part of the transformation of children’s mental health systems to smooth their experience.
The ACA is stable, but 2025 brings uncertainty.
The stability and future success of the ACA marketplaces hinge on the decisions of the 119th Congress regarding the extension of subsidies. If these subsidies are reduced or cancelled it could disrupt what has become a robust and reliable segment of the health insurance market, potentially requiring another pivotal transformation.
Community collaboration can bridge Medicaid health gaps.
New norms are emerging in the Medicaid program. There is unprecedented policy and programmatic work underway to ensure member experiences are informing the design of Medicaid programs as well as the type and pathways for accessing health and health adjacent services. Federal and state government, managed care plans, and providers must work together to bridge the gap to ensure Medicaid programs are best able to serve their members.
Interoperability remains foundational to quality improvement.
We have many different information systems, but when data is pushed to providers to help them manage their patient panel — ED visits, medications, and other data – higher quality care is more likely to be provided to patients.
Provider networks can be structured to ensure success in value.
Medicare providers are embracing value-based care on different timelines and in varied ways. Policymakers, health plans, and other stakeholders need to think carefully about how to structure networks with those providers who are doing it well to get the best results.
Clinics need help with data and financing to drive value-based outcomes.
Poverty is the primary diagnosis for patients of federally qualified health centers (FQHCs), and payment can better recognize the connections FQHCs make to anti-poverty services and programs. As the healthcare industry moves providers along the trajectory of value-based payment, FQHCs will be positioned to deliver whole person care if their data and financing is aligned.
These – and other takeaways and partnerships – will inform strategic, policy, programmatic and operational decisions at the hundreds of organizations represented at the HMA conference. They are also key points as we shape the conversations for the 2025 event.

Webinar Replay: Electoral Consequences: Impact on the ACA Marketplace
This webinar was held on November 20, 2024.
The 2024 elections could create dramatic changes in the ACA marketplace. Enhanced ACA subsidies passed during the pandemic are set to expire in 2025, and a new CMS administrator will shape policy and regulatory components that affect marketplace and consumer dynamics. This webinar is designed for health plans currently participating in the ACA marketplaces, plans who are considering attending, as well as state regulators and marketplace leaders who need to understand changes that might be coming their way. The webinar covered not only what is expected to change (2026 Notice of Benefit and Payment Parameters, AVC) but also what could possibly change that will affect 2025 Marketplaces and beyond.

Webinar Replay: The Future of Medicare Advantage: How the Election Results Impact the Program
This webinar was held on November 19, 2024.
More than 50 percent of Medicare enrollees chose Medicare Advantage (MA) as their preferred coverage option. This growth brought increased scrutiny, with elected officials, regulators, think tanks, and news organizations raising questions about the program’s current makeup. With MA reform potentially on the table in 2025, attendees explored how the election results could impact policy changes in the coming year.
Learning objectives:
- Understand the political landscape for the Medicare Advantage program post-election
- Identify likely Medicare Advantage reforms
- Assess organizational strategies in response to Medicare Advantage reforms

Medicare Advantage Plans: It’s time for the Stars 4th quarter push
The Medicare Stars program is a year-round endeavor for Medicare Advantage (MA) plans. That being said, all MA plans intensify their Stars campaign during the fourth quarter of each year. The most important aspect of the fourth quarter push is to know where to focus. MA Star ratings are more than a quality score—they shape the financial and operational success of MA plans.
Some measures may no longer apply in the fourth quarter. Once the Consumer Assessment of Healthcare Providers & Systems (CAHPS) survey has been completed, the fourth quarter becomes a time to continue the effort to enhance and improve consumer experience for next year’s CAHPS survey. For example, post discharge follow-up is time limited and going back to earlier quarters is not possible. Adherence measures work similarly; if a member has already lost 80% of days covered, coverage cannot be made up during the fourth quarter. The message should be “focus only on measures where you can make a difference.”
Mammograms and colorectal exams can occur any time of year. These two measures should definitely be the focus of a fourth quarter push. A constraint may be provider capacity since all MA plans are focused on the same measures. Measures with low denominators like the osteoporosis management measure may be an important element in fourth quarter strategies.
Star Ratings and Operational Excellence
Operational excellence begins with robust, accurate, and actionable data, and even using lean six sigma principles to drive process improvement. Advanced analytics platforms are needed to aggregate and analyze vast amounts of healthcare data and operational data. Accurate risk adjustments, quality measurement, and operational metrics like appeals are essential. Data discrepancies or delays result in penalties, lower Star ratings, and incorrect payments. MA plans must develop processes to validate via quality assurance process and audit data regularly against CMS requirements.
Operational excellence also relies on a well-trained engaged workforce. Training should focus on fostering a culture of continuous improvement, where every team member is aligned with the organization’s goals of improving quality and operational performance.
Star ratings are a byproduct of strategic, data-driven approaches to care management, quality improvement, and operational efficiency. Success lies in the ability to optimize data integrity, streamline care coordination, and proactively resolve member concerns.
Accelerating Star Rating Performance
The HMA Stars Accelerator Solution offers a comprehensive, results-oriented approach to Star Rating performance improvement that addresses the multifaceted challenges faced by health plans and makes sure that your intensified 4th quarter effort is productive. The HMA Stars Accelerator Solution analytics provides information to plans about prioritizing measures during the fourth quarter push. It examines your plans leadership structure, operational processes, technology, reporting, member-centric engagement, provider partnerships, and develops a strategy for your organization using a data-driven approach for continuous improvement. Multiple “what-if” scenarios are developed that identify top priorities. Measure thresholds that are too far to reach are replaced by measures that are within reach during the final months of the year. The Accelerator approach includes “all-hands-on-deck” – care coordination, customer service, network development, marketing, analytics, and others. Accelerator plans introduce provider and member incentives and/or fee schedule adjustments to increase interest. These plans also provide information to providers on those attributed members who have measure gaps to facilitate provider outreach that is coordinated with plan outreach.
As the fourth quarter push occurs in the middle of the Annual Enrollment Period, lessons learned can be applied immediately. The HMA team can backstop your organization during this very busy time, avoiding missed opportunities. The fourth quarter push does not end until midnight January 31st of each year.
The HMA Stars Accelerator Solution will create a permanent change in your organization that is designed to yield a 4-star rating or higher each year. The Accelerator is a cultural transformation designed to strengthen star performance. Click here to learn more about the HMA Stars Accelerator Solution’s capabilities, where you can request a copy of the HMA Stars Accelerator Playbook. Let’s have a conversation about how your fourth quarter push is designed and unfolding.
We are also holding two webinars that may be of interest:
Falling Stars: Who’s Who in the 2025 Star Ratings
November 7, 2024 – 3:30 PM ET
Register now
Colleagues from Wakely Consulting Group, an HMA Company, will discuss trends in Overall Star Ratings, the appeals and lawsuits filed in response, and future changes to the Star Rating program that are likely to depress Star Ratings even further over the next few years.
Mastering Star Performance: Strategies from the HMA Stars Accelerator Program
November 13, 2024 – 12:00 PM ET
Register now

CMS releases draft benefit and payment parameters for 2026 Marketplace
Our second In Focus article reviews the recently proposed Notice of Benefit and Payment Parameters (NBPP) for 2026. The Centers for Medicare & Medicaid Services (CMS) proposed rule, released October 10, 2024, describes the policy and payment changes that will affect the Affordable Care Act (ACA) markets in 2026. Public comments must be submitted to CMS by November 12, 2024. Key highlights from the proposed rule follow.
Broker Oversight and Monitoring
CMS proposes to increase oversight and accountability for brokers and agents that write policies through HealthCare.gov. In response to the discovery earlier this year of fraudulent actors reassigning broker designations and switching consumer enrollments without their permission or knowledge, CMS has already implemented several corrective actions, including the suspension of 850 Healthcare.gov agents and brokers. CMS intends to build on these actions through the following interventions:
- Clarify that lead agents, typically an agency owner or executive, are subject to the same rules as individual brokers, agents, and web-brokers and that enforcement action can be taken against the lead agents if they explicitly or implicitly condone misconduct or fraud
- Broaden CMS’s authority to suspend broker and agent system access, inclusive of instances of suspected misconduct that affects eligibility determinations, operations, applicants, or systems
- Update the model consent form to include documentation of the broker reviewing and confirming the accuracy of submitted application information with the consumer.
Marketplace User Fees
CMS proposes to increase the user fee collected to pay for administration of HealthCare.gov as follows:
- Between 1.8 percent and 2.5 percent in 2026 for federally facilitated marketplaces (FFM) states, up from 1.5 percent of monthly premiums in 2025
- Between 1.4 percent and 2 percent in 2026 for state-based marketplaces on the federal platform (SBM-FPs), up from 1.2 percent in 2025
The proposed changes are due, in part, to uncertainty caused by the future of the enhanced premium tax credits that are set to expire at the end of 2025. The enhanced premium tax credits are the driving force behind the increase in nationwide marketplace enrollment to more than 21 million people in 2020 from 11.4 million in 2020. If not extended, or if it takes past March 2025 for Congress to act, CMS has indicated the user fees will increase in 2026 to 2.5 percent for FFM states and 2% for SBM-FPs to accommodate expected enrollment declines. Notably, after several years of significant decreases, CMS is proposing to increase the user fees above 2025 levels regardless of the outcome of the enhanced premium tax credits.
Plan Limits for Non-Standard Plans
CMS proposes to clarify rules limiting the number of non-standardized plans an issuer can offer through HealthCare.gov (two or less in 2025). The limit is applied per product network type (e.g., HMO, PPO), per metal level, per service area, per inclusion of adult/pediatric dental and/or vision benefits (with additional exceptions, starting in payment year (PY) 2025, for plans with specific design features that would substantially benefit consumers and meet other requirements). To maximize the number of non-standardized plans offered on HealthCare.gov, an issuer could offer up to 16 plans per metal level and network type in a given service area by creating every combination of adult dental, pediatric dental, and adult vision (or even more, if plans meet the exception requirements).
Though CMS does not limit the number of standardized plan options an issuer offers on HealthCare.gov, they propose reinstating a meaningful difference standard to prevent consumer confusion and unnecessary plan proliferation. The proposed standard is similar to the removed standard from 2019; for plans in the same metal level, product type, and service area, a reasonable consumer needs to be able to identify at least one material difference in benefit coverage, provider networks, and/or formulary.
New Premium Payment Threshold Options for Issuers
CMS proposes new options for issuers to avoid triggering late payment grace periods for consumers who make most but not all of their premium payment to minimize termination of coverage for consumers who owe a small amount. The options include:
- The current option of a “reasonable” percentage of net premium. CMS proposes codifying 95 percent as the minimum threshold.
- New proposals of as low as 99 percent of gross premium and a fixed-dollar threshold of $5 or less.
CMS is also considering limiting issuers to offering just one payment threshold option—either fixed-dollar or percent of premium—to avoid consumer confusion.
Increased Transparency for State-Based Marketplaces
CMS proposes new initiatives to promote transparency into state-based marketplace (SBM) program operations. These initiatives include:
- Publishing State Marketplace Annual Report Tool (SMART) submissions, which are used to monitor SBM compliance with select eligibility and enrollment, program integrity, and financial reporting requirements. SBMs must annually participate in independent programmatic and financial audits as part of SMART. CMS proposes to make the 2023 SMART submissions public in spring 2025.
- Expanding the disclosure of SBM information to include data collected but not currently published, including details on SBM eligibility, enrollment, and plan certification policies as well as Navigator program spending, call center metrics, and website traffic data.
SBMs already are required to publish programmatic and financial audit summaries and generally publish robust data and information on their program operations through public reports and meetings; however, this information is neither centrally located nor consistently published across all SBMs.
Key Considerations
The proposed 2026 NBPP would build on previous actions that CMS has taken to address fraudulent broker and agent activity and to shore up financial sustainability of Healthcare.gov operations in light of uncertainty about the enhanced premium tax credits. It also seeks to make clear how plan variations adding dental or vision benefits factor into HealthCare.gov plan limits and gives issuers new premium payment threshold options. Lastly, it proposes new transparency requirements for SBMs. Interested stakeholders, including SBMs and issuers, should monitor how these proposed changes will affect consumers, operational processes, product strategy, and financial sustainability.
Connect With Us
The Health Management Associates, Inc., team has the depth, experience, and subject matter expertise to assist with tailored analysis and the modeling capabilities to assess the policy impacts to consumers, marketplaces, and issuers. If you have questions or want to discuss the proposed rule, contact our featured experts below.
For additional information on elements of the proposed NBPP not discussed here, Wakely Consulting’s white paper, Summary of Provisions of HHS’ Proposed 2026 Notice of Benefit and Payment Parameters and Other Key Regulations, highlights the proposed changes to the Risk Adjustment program, Medical Loss Ratio, and the Actuarial Value Calculator, among other changes.
